Quantification of ginkgolides and bilobalide by HPTLC
In addition to the identification, this case study demonstrates how ginkgolides and bilobalide in Ginkgo biloba extracts can be quantified by HPTLC.
Introduction
HPTLC is the only single test that allows for 100% botanical ingredient identification. HPTLC is the primary chemical test for identification of all pharmacopoeia monographs on botanicals. According corresponding monographs of USP, PhEur and AHP on Ginkgo leaf and Ginkgo leaf extract are identified by HPTLC based on fingerprints of flavonoids. In addition to the identification, our case study method demonstrates how ginkgolides and bilobalide in Ginkgo biloba extracts can be quantified by HPTLC.
Challenge
Most techniques can address identification only in part. They either are limited to plant parts (microscopy), have difficulties with natural variability of botanical materials (IR techniques), or focus on quantitative comparison of separated markers (HPLC). All other techniques lack the versatility and flexibility of HPTLC for the analysis of botanicals. HPTLC allows for convenient comparison of many samples side by side, where similarities and differences can be clearly seen. Quantification of the separated compounds is possible by densitometry using our TLC Scanner 4.
Description of the procedure documentation
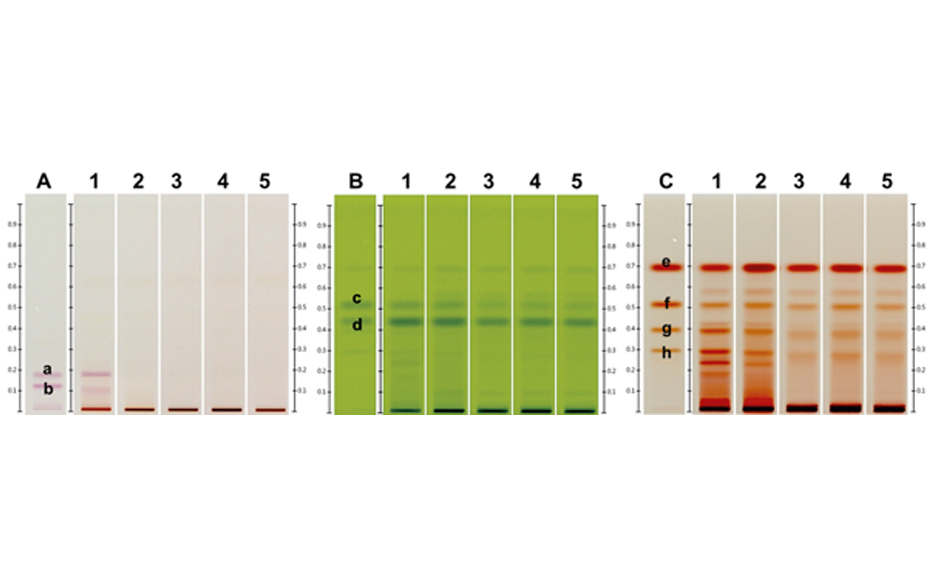
Ginkgo extracts and the standards are prepared according to CAMAG’s standardized method (A-92.1). The HPTLC plate is impregnated with sodium acetate. All parameters (conditions for sample application, chromatogram development, and evaluation) are logged into the winCATS software which controls all major CAMAG instruments needed for the HPTLC analysis. The samples and standards are applied bandwise on the impregnated HPTLC plate using the ATS 4, which guarantees precise and reliable application. Then the HPTLC plate is developed under standardized conditions in a saturated chamber using the ADC 2. The ADC 2 is unsurpassed for reproducibility since all developing parameters are controlled from plate to plate, regardless of environment. The developed HPTLC plate is evaluated and documented by the TLC Visualizer in three modes, white light, UV 254 nm, and UV 366 nm. By the use of HPTLC plates containing a fluorescence indicator, all analytes which absorb UV 254 nm can be detected prior to derivatization. Derivatization increases the specificity and selectivity of the method. For the analysis of ginkgolides and bilobalide in Ginkgo extracts the plate is sprayed evenly with acetic anhydride, heated at 180°C, and documented by the TLC Visualizer under UV 366 nm. For quantification each track is scanned in absorption at 300 nm with the TLC Scanner 4.
-
01
Standard mix: 1, 2, 5, 7 and 10 μL applied (image after derivatization, under UV 366 nm)
-
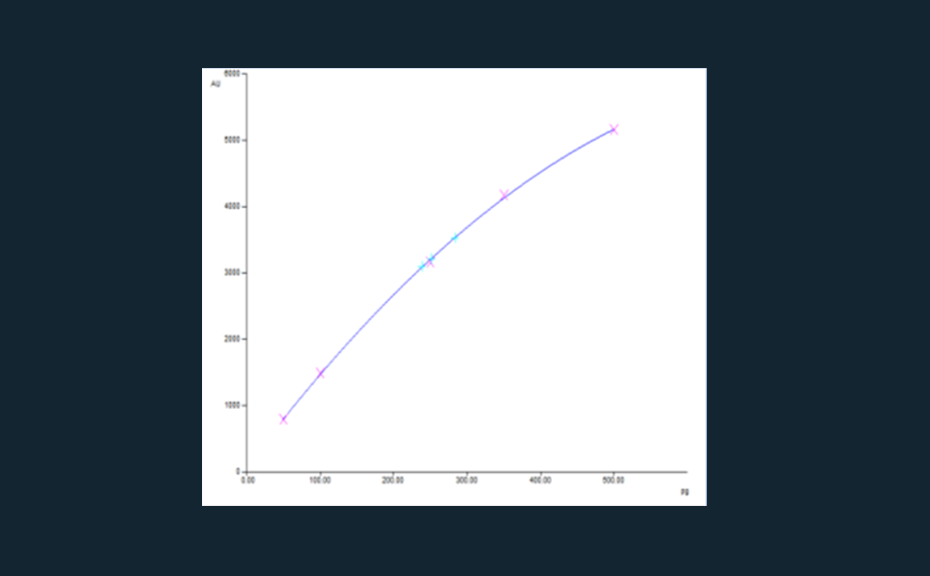
02
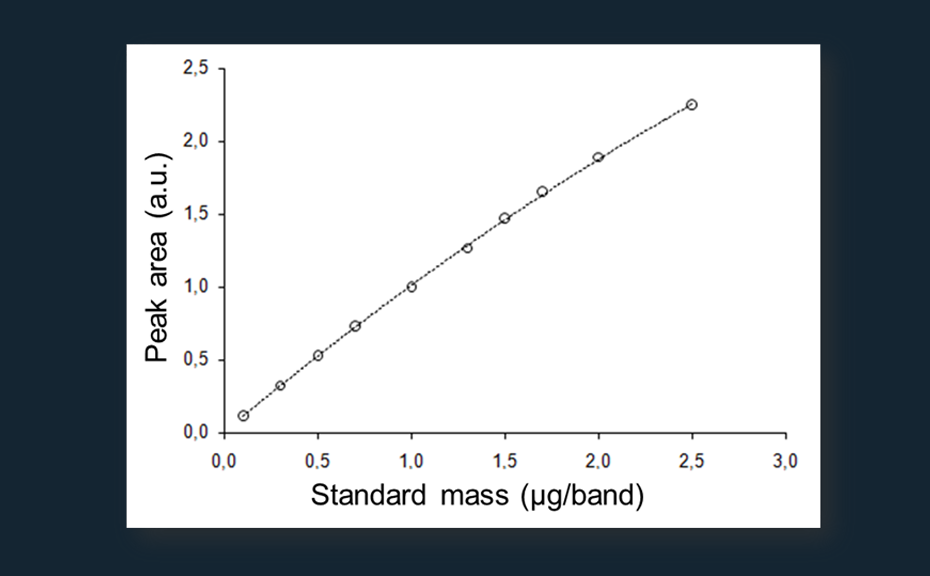
Calibration curve of ginkgolide B (pink: standards, turquoise: samples, r=0.9998, sdv: 1.56%
-
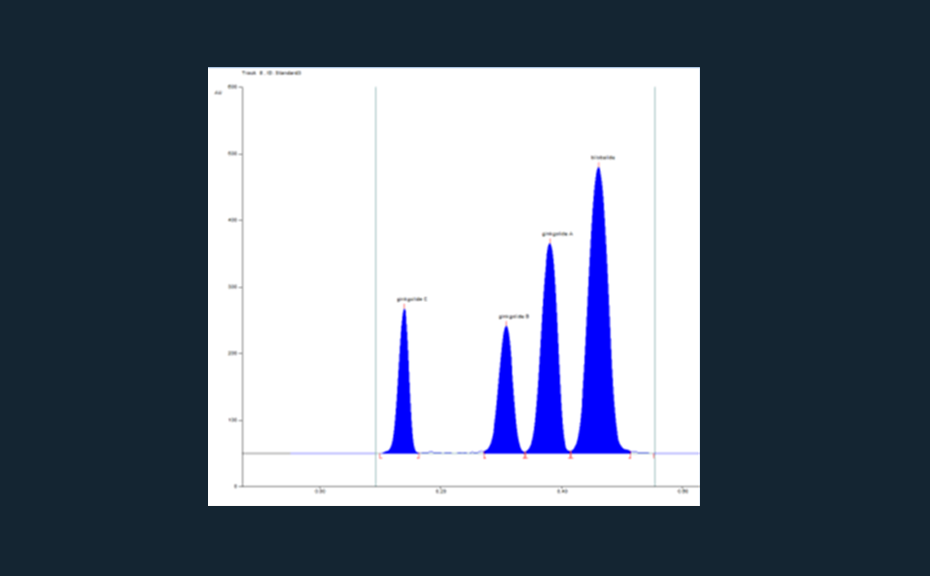
03
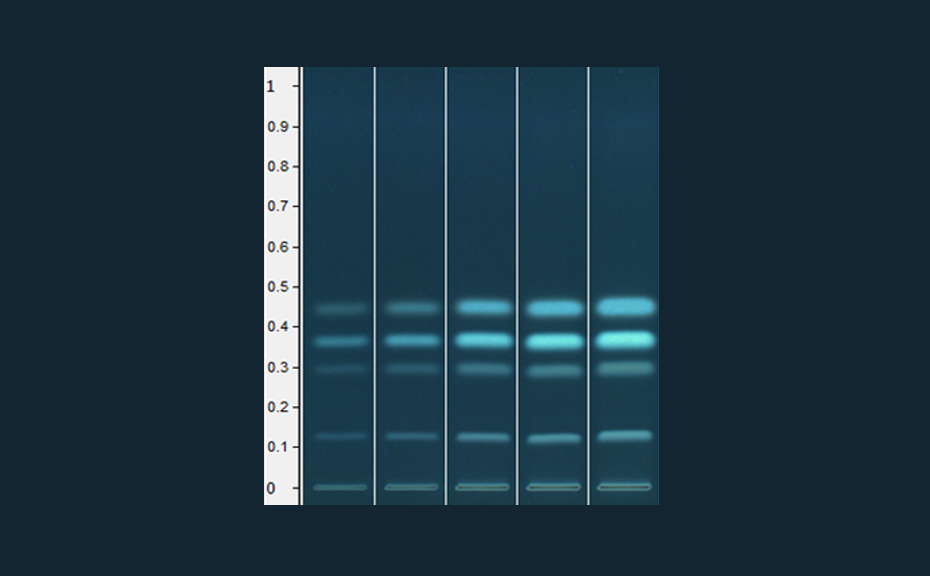
Chromatogram of the separated standards (ginkoglide C, ginkoglide B, ginkoglide A, and bilobalide with increasing RF value, 5 μL applied)
-
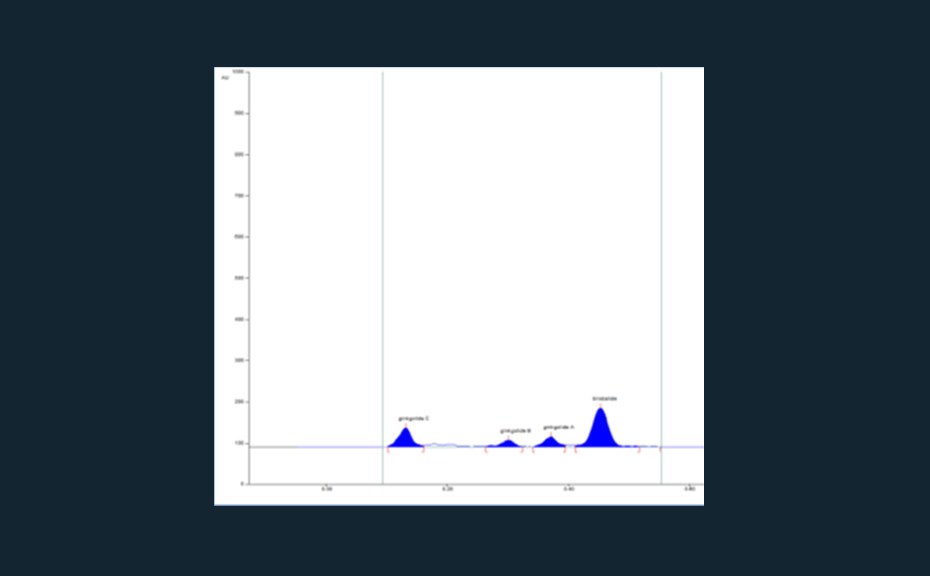
04
Chromatogram of the adulterated sample (Ginkgo leaf extract mixed with cellulose, 5 μL applied)
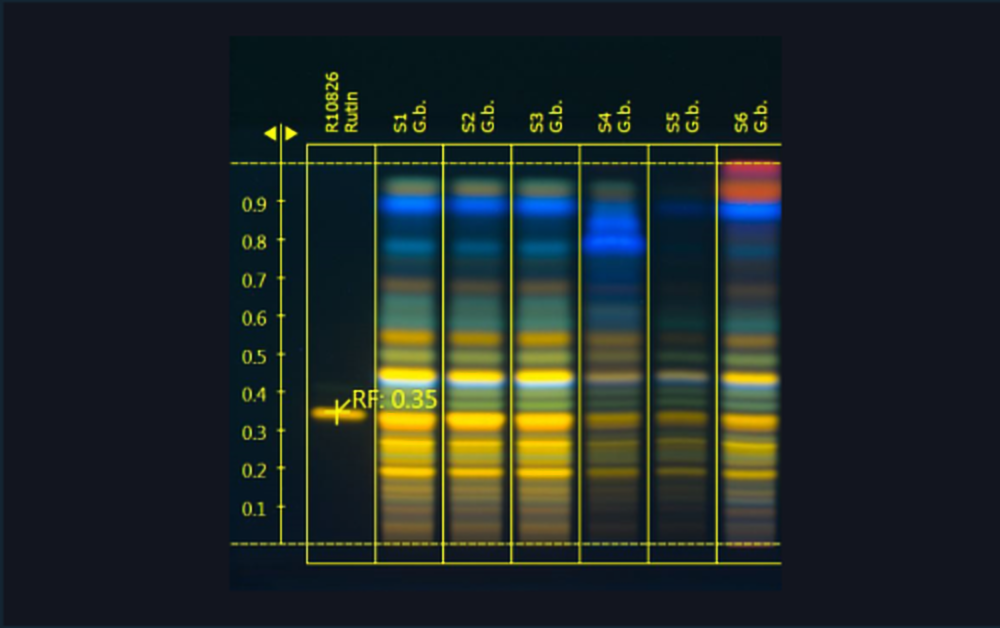
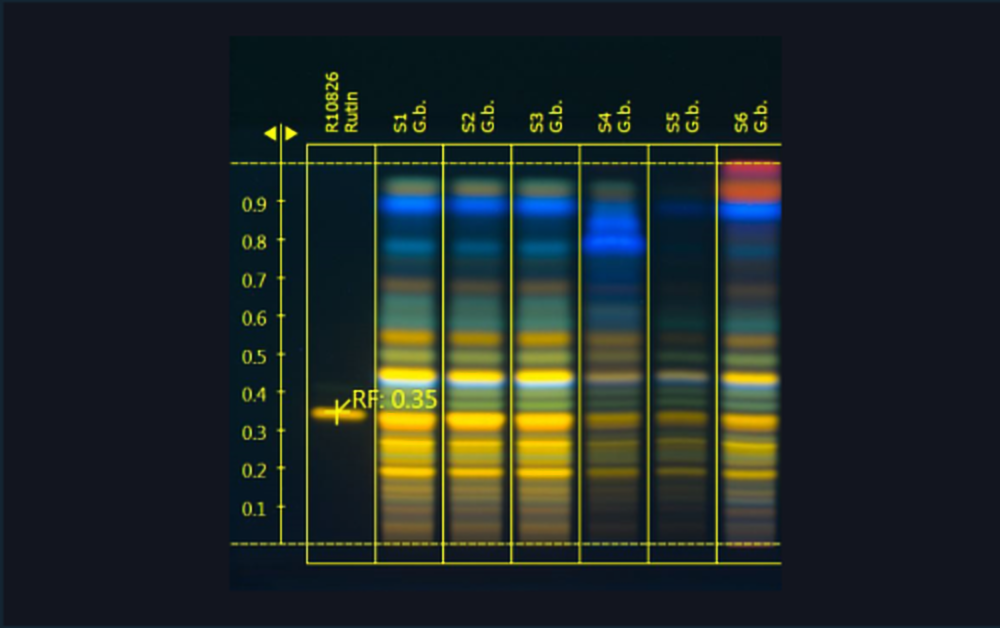
| 1 | Ginkgo leaf extract (*BRM), 5 µL |
| 2 | Standard mix (bilobalide, ginkgolides A, B, and C), 1 µL |
| 3 | Ginkgo leaf extract, 5 µL |
| 4 | Standard mix (bilobalide, ginkgolides A, B, and C), 2 µL |
| 5 | Ginkgo leaf extract, 5 µL |
| 6 | Standard mix (bilobalide, ginkgolides A, B, and C), 5 µL |
| 7 | Ginkgo leaf extract mixed with green tea (additional blue bands in the upper zone of the chromatogram), 5 µL |
| 8 | Standard mix (bilobalide, ginkgolides A, B, and C), 7 µL |
| 9 | Ginkgo leaf extract mixed with cellulose (less intensity of all flavonoids), 5 µL |
| 10 | Standard mix (bilobalide, ginkgolides A, B, and C), 10 µL |
Benefits of using modern HPTLC for the analysis of herbal medicines and botanical dietary supplements
- HPTLC shows visually the similarities and differences between samples and references.
- For the identification of herbal drugs and other naturally derived materials standardized HPTLC is the method of choice and recommended by pharmacopoeias worldwide.
- By using internal or external chemical reference standards and the TLC Scanner 4 marker compounds can be quantified precisely.
- HPTLC can also be used for assays (potency), purity (adulteration), and stability studies (shelf life).
- The whole sample is detectable on the plate and allows to judge all components of the sample even if components remain at the application zone.
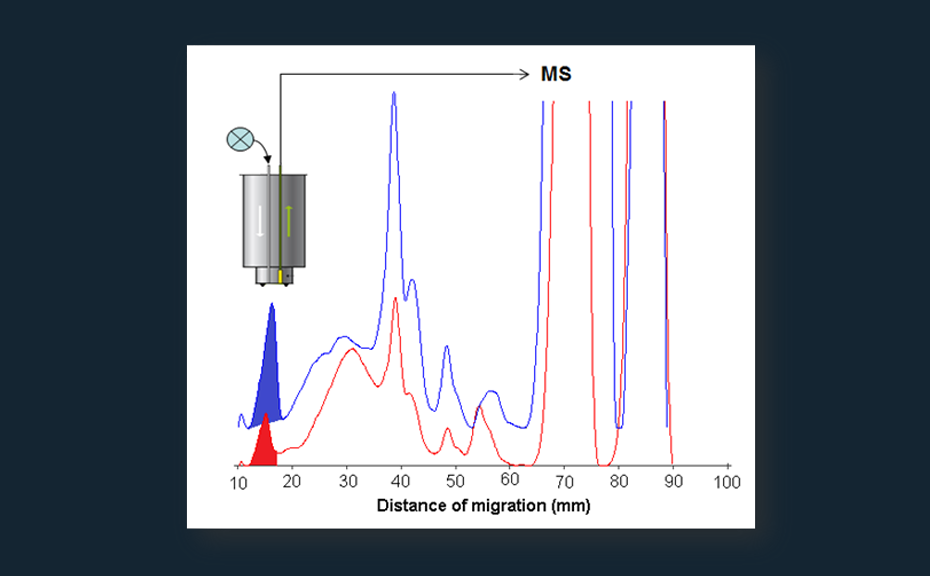
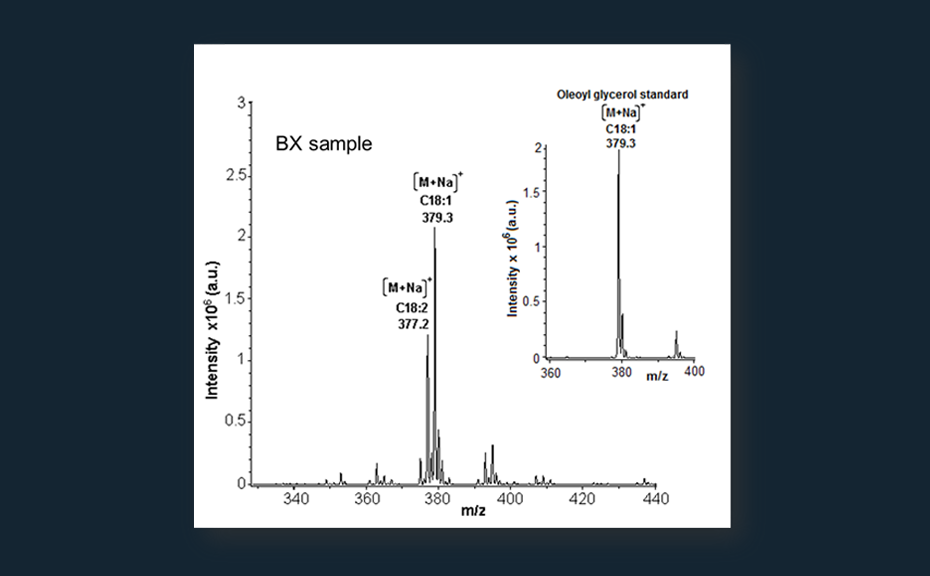
- Following separation and localization of target zones by non-destructive detection analytes can be directly eluted from the plates with the TLC-MS Interface and identified by hyphenated techniques (e.g. MS, NIR, NMR).




![HPTLC-MS full scan mass spectrum of eluted estrogen active zone with the expected mass m/z 339 [M-H]-](/wp-content/uploads/2024/07/cs_estrogens_img_2.png)