Standardized HPTLC for Reproducible Chemical Fingerprinting
Methods in Molecular Biology, 2025
Authors: Thi Kieu Tiên Do, Ilona Trettin & Eike Reich
Methods in Molecular Biology, 2025
Authors: Thi Kieu Tiên Do, Ilona Trettin & Eike Reich
Nammex specializes in the production of high-quality, certified organic mushroom extract powders for the food and dietary supplement (DS) industries. As a result of the rapid growth of the functional mushroom market, we have observed the introduction of many new products of varying quality. Nammex has a long-standing history of leading the industry in product analysis, with a focus on ensuring product authenticity and efficacy [1]. Our laboratory has developed an innovative HPTLC method for the identification and quality control testing of diverse species used in DS products. With this method, we aim to enhance the overall reliability and transparency of quality testing in the industry.
The functional mushroom market is experiencing significant growth, driven by factors like increased DS usage and ongoing medical research. Despite the market’s size, only one validated HPTLC mushroom identification method has been published (USP Ganoderma lucidum monograph) [2], and its indiscriminate use across other species may lead to misidentification, undermining the reliability of the identification process and creating a need for more comprehensive testing solutions.
HPTLC is widely recognized for its effectiveness in botanical identification, making it an ideal method for mushroom analysis. In the absence of validated methods, consumers risk exposure to mislabeled or adulterated products. For instance, products containing tempeh-like mycelium (i.e. vegetative body) fermented grain are often marketed as mushrooms (i.e. fruiting bodies) despite significant compositional differences. Additionally, concentrated mushroom extracts may be deficient in specific marker compounds due to processing conditions.
HPTLC offers a robust, highly selective approach for mushroom differentiation. This new method ensures that characteristic compounds from diverse chemical classes in mushrooms are clearly separated, supporting accurate species identification. The advantages of HPTLC in this context include its specificity, versatility, and ability to detect adulteration in complex products.
Standard solutions
Standard stock solutions are prepared at 0.5 mg/mL in methanol.
Samples consist of 250 mg of mushroom extract powder or finely milled whole mushrooms. These are extracted in 5.0 mL of methanol, vortexed for 10 s, sonicated for 10 min at room temperature, and centrifuged at 3500 rpm for 10 min. The supernatant is then transferred to vials.
HPTLC plates silica gel 60 F254 Premium Purity (Supelco, Merck), 20 × 10 cm are used.
10.0 μL of sample solutions and 2.0 μL of standard solutions are applied as bands with the Automatic TLC Sampler (ATS 4), 15 tracks, band length 8.0 mm, distance from the left edge 20.0 mm, track distance 11.4 mm, distance from the lower edge 8.0 mm.
Plates are developed in the ADC 2, with chamber saturation (with filter paper) for 20 min and after activation at 33 % relative humidity for 10 min using a saturated magnesium chloride solution, development with toluene – methanol – and acetic acid 85:10:5 (V/V) to the migration distance of 70 mm (from the lower edge), followed by drying for 5 min.
Post-chromatographic derivatization
The plates are immersed into p-anisaldehyde sulfuric acid reagent using the Chromatogram Immersion Device (immersion speed: 5 cm/s, immersion time: 0 s). After derivatization, the plates are heated at 100 °C for 4 min using the TLC Plate Heater.
Documentation
Images of the plates are captured with the TLC Visualizer 3 in UV 254 nm, UV 366 nm, and white light after development, and again after derivatization in UV 366 nm and white light.
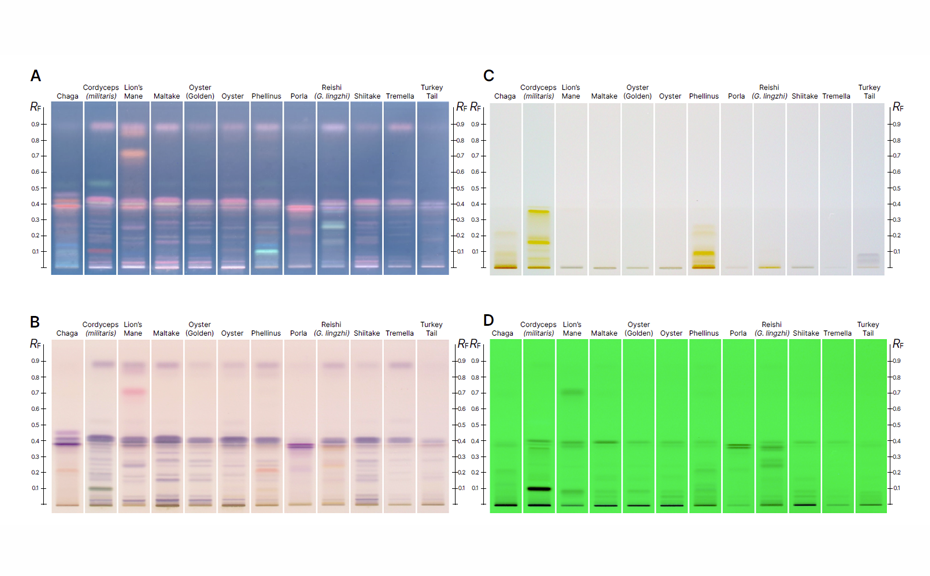
The high selectivity of the HPTLC method is demonstrated through distinct chromatographic fingerprints obtained for each species. These fingerprints display characteristic bands under multiple detection modes, providing a reliable means of differentiating between species and product types such as mushroom extracts and mycelia fermented grain powders.
Key marker compounds for each species were identified through literature, playing a critical role in distinguishing between the mushroom and the mycelium. Specifically, the mushroom is known to exhibit a different profile of compounds than the mycelium. HPTLC comparisons of mushroom extracts, supported by these chemical markers, effectively demonstrate these differences.
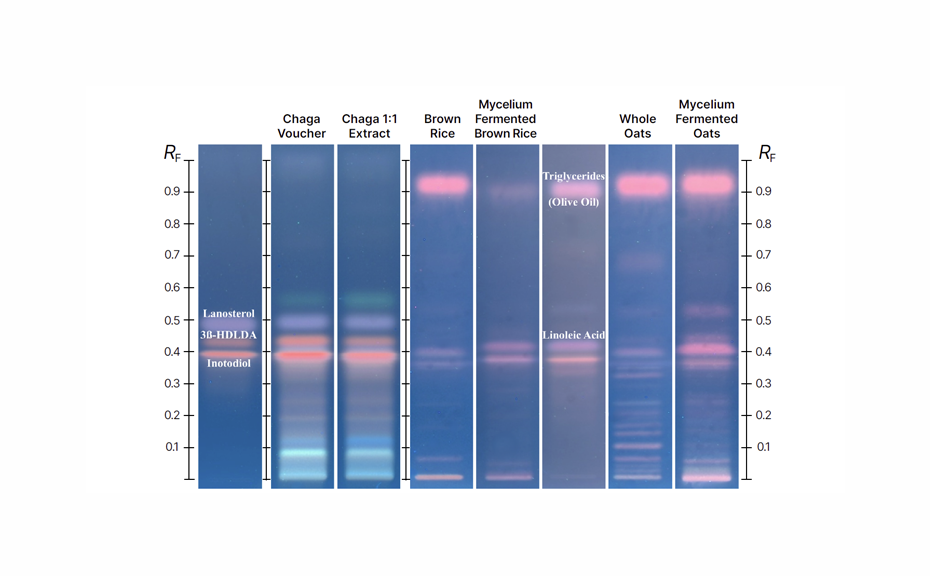
HPTLC comparisons between Chaga conk, pure mycelium, and fermented grain forms reveal significant compositional differences, with fermented grain fingerprints closely matching grain reference materials. Importantly, Chaga triterpenoid markers are absent in fermented grain, which instead shows high concentrations of triglycerides and linoleic acid. These chromatograms highlight the clear differences between Chaga conk, 1:1 extract, brown rice and oats, and fermented grain products, underscoring HPTLC’s effectiveness in detecting potential adulteration and verifying product authenticity.
While fermented grain products are expected to contain grain, the lack of sufficient mycelium or relevant compounds, along with unclear labeling practices, raises concerns about product authenticity. Many fermented grain products prominently display “mushroom” on the front label, along with images of mushrooms, but only disclose their myceliated grain content on the back, with some brands failing to identify the grain entirely. This inconsistency in labeling, coupled with the compositional differences identified through HPTLC, underscores the urgent need for more transparent and stringent quality control measures in the mushroom supplement industry.
01
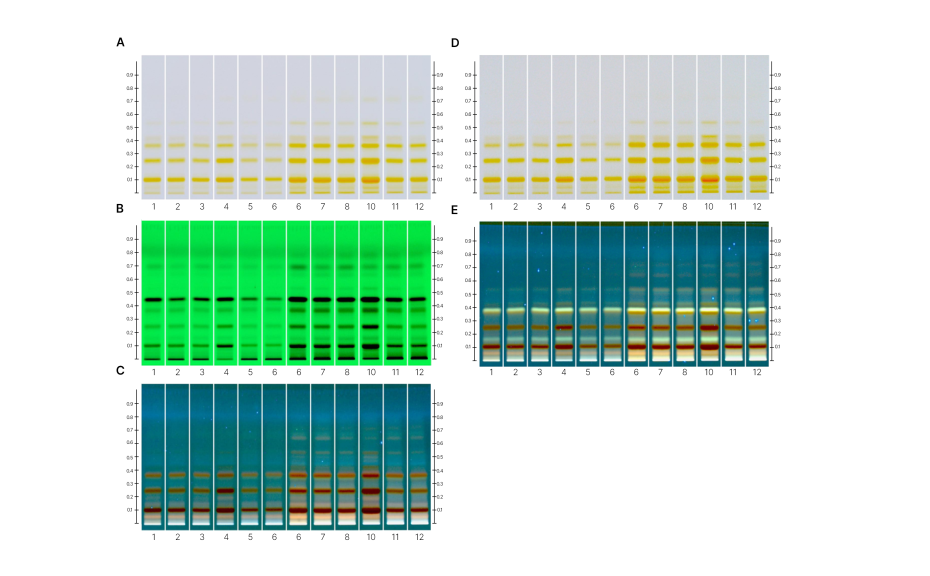
HPTLC chromatograms of whole mushroom, conk, or sclerotium vouchers from 12 species, highlighting compositional differences between species under various detection modes. Images after derivatization are shown in UV 366 nm (A) and white light (B). Chromatograms captured after development are displayed in white light (C) and 254 nm UV light (D).
02
HPTLC comparisons between Chaga conk voucher and fermented grain forms reveal significant compositional differences, with fermented grain fingerprints closely matching grain reference materials. Key Chaga marker compounds – such as inotodiol (
In conclusion, the development of the innovative HPTLC method for the differentiation of functional mushrooms offers a significant advancement in ensuring product authenticity and quality within the growing mushroom supplement market. By providing clear, reliable chromatographic fingerprints for various species, this method enhances the ability to detect adulteration and verify product composition, particularly in distinguishing between mycelia fermented grain-based products marketed as mushrooms. As the market continues to expand, the implementation of robust, transparent quality control measures like HPTLC will be critical in maintaining consumer trust and safeguarding product efficacy.
Literature
[1] Chilton, Jeff. White Paper. Redefining Medicinal Mushrooms: A New Scientific Screening Program for Active Compounds. Nammex, 2015. jeff@nammex.com
[2] United States Pharmacopeia (USP). Ganoderma lucidum Fruiting Body Monograph. USP 43-NF 38, United States Pharmacopeial Convention, Rockville, MD, 2020.
Contact: Coleton Windsor, Nammex, Box 1780, Gibsons, British Columbia, Canada, coleton@nammex.com
For the past 20 years, CAMAG Laboratory has been a key contributor to pharmacopoeias worldwide, developing identification methods for botanicals, herbal drugs, and extracts. As a pioneer in standard-setting efforts, Dr. Eike Reich played a crucial role as an HPTLC expert in numerous pharmacopoeia committees. Now, as he transitions into retirement, Dr. Reich passes the torch to Dr. Tiên Do and her team, who continue to advance this important work.
To effectively support the pharmacopoeia committees, all members of the laboratory undergo extensive training in working with standardized methodologies. Delivering HPTLC methods tailored to the specific requirements of pharmacopoeias involves more than just standardized HPTLC; each scientist must also understand and follow a general method development process. This process encompasses several key stages, illustrated in this paper using the European Pharmacopoeia (Ph. Eur.) monograph on Epimedium leaf as an example.
As the preferred chromatographic technique for the identification of herbal drugs, HPTLC aims to determine a characteristic chromatogram (fingerprint) based on the relative position, color, and intensity of specific zones. According to Ph. Eur., HPTLC must adhere to the Ph. Eur.’s general chapter 2.8.25., which specifies all steps and parameters of the HPTLC process. This document describes in detail the specific points relevant to the development of an HPTLC identification method.
Developing a suitable identification method involves several steps:
Step 1: definition
The scope of the method must clearly specify the article (e.g. the medicinal plant) to be identified. In addition to the Latin plant name, the definition should include the accepted plant part(s) and the process by which the article is obtained (drying, cutting, extracting, etc.). Ideally, an identification method is specific for the article of the monograph and distinguishes related articles that may be considered adulterants.
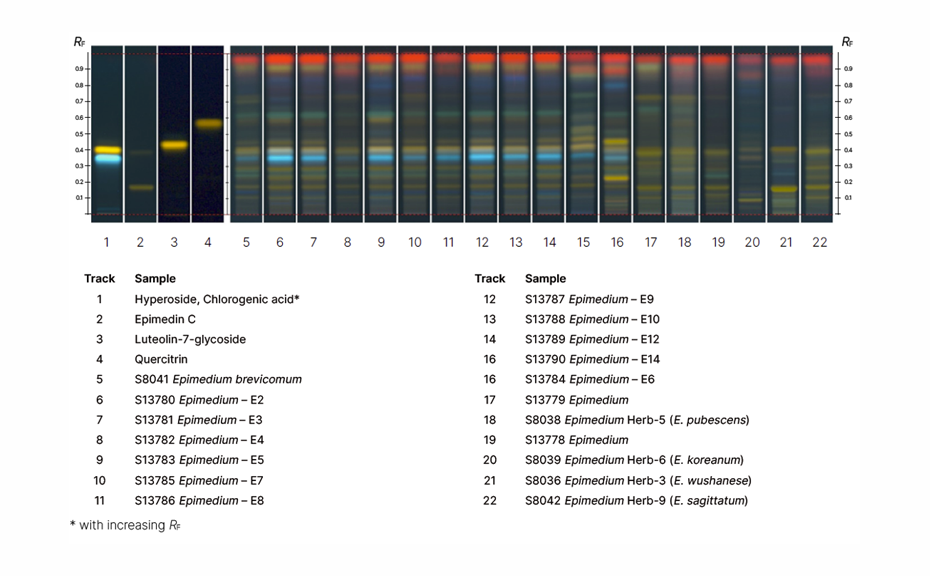
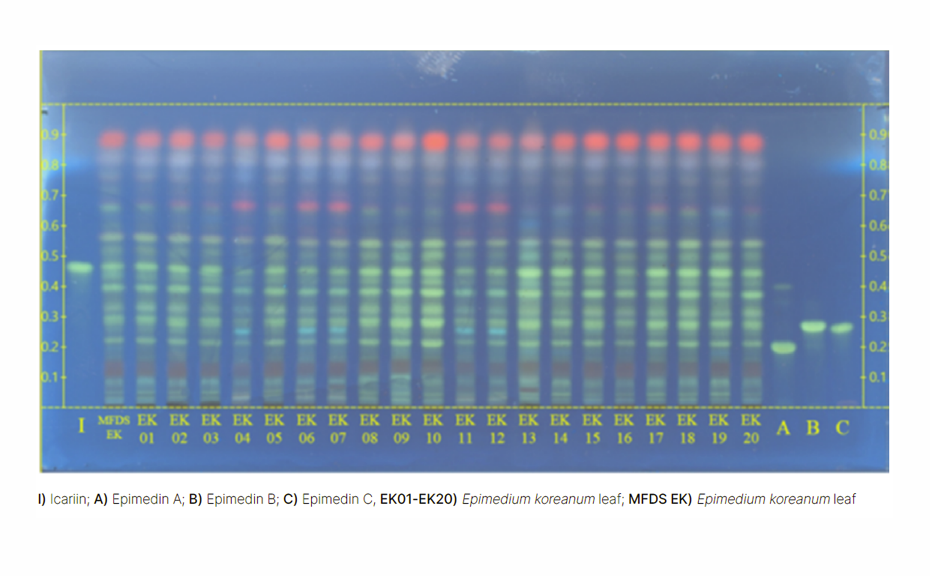
Various monographs on “Epimedium” target the whole or fragmented dried leaf or herb of several species (see table) according to availability in different markets. The Ph. Eur. monograph on Epimedium leaf includes whole or fragmented dried leaf of the major species E. koreanum Nakai, E. brevicornum Maxim., and E. pubescens Maxim., including mixtures thereof.
Acceptance criteria for the herbal drug “Epimedium leaf” must include the selected drugs and exclude all others (e.g. E. sagittatum).
Step 2: collection of samples
Samples of different origins and related species are collected by the pharmacopeia group and distributed to various collaborating laboratories. Each laboratory also collects its own samples. A wide range of samples is crucial to ensure that the method is applicable to routine analysis of market samples.
Step 3: development / evaluation of HPTLC method(s)
Using standard HPTLC conditions, methods from pharmacopoeias are evaluated for reproducibility, practicality, and fitness for purpose. Other methods can also be considered. For Epimedium leaf, several methods have been proposed, each with specific advantages and limitations.
A first proposal was made to the Traditional Chinese Medicine (TCM) Working Party by the Shanghai Institute for Materia Medica (SIMM), using water, formic acid, n-butanol, ethyl acetate 1:1:3:6 (V/V/V/V) as developing solvent. During the peer review in our laboratory, the RF values were lower and the colors of zones slightly different.
This prompted us to optimize sample preparation, developing solvents, and detection, based on a previously established method for separation of flavonoids, using ethyl acetate – formic acid – water 8:1:1 (V/V) and derivatization with NP/PEG reagents.
SMOOTH & PRECISE OPERATION
01
Evaluation of the first proposal
02
Second proposal
In parallel, a third method with good reproducibility, using ethanol – ethyl acetate – water 2:1:8 (V/V) was developed for consideration by the United States Pharmacopoeia by the Korean group led by Prof. Jang (Kyung Hee University). For compliance with Ph. Eur. Chapter 2.8.25, we included a System Suitability Test (SST) and intensity markers.
Third proposal
Third proposal
Step 4: method selection and acceptance criteria definiton
In several iterations, the experts compare the submitted proposals and reach agreement on the most suitable one. With this method, multiple samples are analysed, and the results are described in table format. The data is included in the monograph and published for public comment. In the case of Epimedium leaf, species can be clearly discriminated. The result table describes only the features common to the species covered by the monograph.
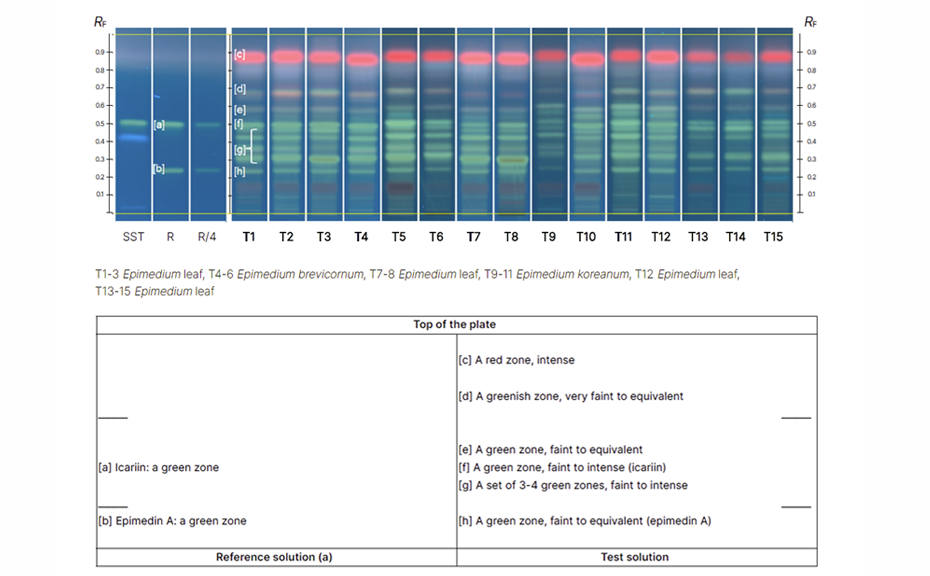
Data included in the monograph and published for public comment
Data included in the monograph and published for public comment
Step 5: public comments and finalization of method
Comments received from various stakeholders are reviewed by the expert committee before the monograph is presented to the pharmacopoeia commission for adoption. After publication in the Ph. Eur., the HPTLC fingerprints are shown in the EDQM knowledge database.
For CAMAG Laboratory, the involvement in the development and refinement of HPTLC methods not only contributes to global pharmacopoeia standards but also strengthens the scientific rigor and consistency in the identification of herbal drugs. The ongoing collaboration with international groups ensures that these methods are both practical and scientifically sound.
Contact: Dr. Tiên Do, Sonnenmattstrasse 11, 4132 Muttenz, Switzerland, tien.do@camag.com
The evaluation of edible oil authenticity has become increasingly important due to rising incidents of oil adulteration, where low-quality or non-edible oils are mixed with premium oils for economic gain. Such fraudulent practices not only erode consumer trust but also pose health risks. As adulteration methods become more sophisticated, reliable and efficient detection methods are needed.
This study evaluates the use of HPTLC as a cost-effective and efficient tool for monitoring oil authenticity. Both untargeted (fingerprint profiling) and targeted (mineral oil detection) methods were applied to palm, sunflower, and rapeseed oils, demonstrating the capability to detect adulteration at levels between 5% and 25%.
HPTLC offers numerous advantages, including the ability to analyze multiple samples simultaneously with lower solvent consumption. It is also adaptable to different detection protocols and highly reproducible across laboratories. As a result, HPTLC is positioned as an ideal method for industrial applications requiring rapid and user-friendly solutions for oil quality monitoring.
HPTLC silica gel 60 F254 plates (Merck) were used for vegetable oil analysis, while RP18 F254 plates (Merck) were employed for mineral oil adulteration detection. For mineral oil method, the plates were prewashed with methanol and heated at 110 °C for 15 minutes before application.
Oil samples were applied as 6.0 mm bands onto the plates using an Automatic TLC Sampler 4.
Plates were developed in the ADC 2 to a migration distance of 70 mm for edible oils and 30 mm for mineral oil detection. A mixture of acetonitrile and CPME (7:3 V/V) was used as the developing solvent for vegetable oils, and cyclohexane was used for mineral oil detection. Relative humidity was adjusted to 33% for 10 minutes only for the edible oil method, and chamber saturation was maintained for 20 minutes for both methods.
Post-chromatographic derivatization
After development, chemical derivatization was performed using anisaldehyde reagent for edible oils and primuline reagent for mineral oils. The plates were sprayed with the respective derivatization reagent using the Derivatizer. In the case of anisaldehyde reagent the plates were heated at 100 °C for 3 minutes, and after primuline at 40 °C for 3 min.
Documentation
The plates were documented using the TLC Visualizer 2 at UV 366 nm for mineral oils after derivatization with primuline, and in white light (transmission) for edible oils after derivatization with anisaldehyde reagent. Peak profiles from images (PPIs) were analyzed with the visionCATS software, and peak heights were recorded to assess the presence of adulterants.
Statistical analysis was conducted to assess batch variability and adulteration detection. The peak heights from RF values ranging between 0.2 and 0.8 were used to evaluate oil authenticity. The detection limit for adulteration was established at 5% for both edible oils and mineral oils.
The results demonstrate the successful application of HPTLC in detecting adulteration in edible oils. The method provided clear and reproducible chromatographic fingerprints for sunflower, rapeseed, and palm oils. Each oil type exhibited unique RF values, enabling the differentiation of authentic oils from adulterated ones.
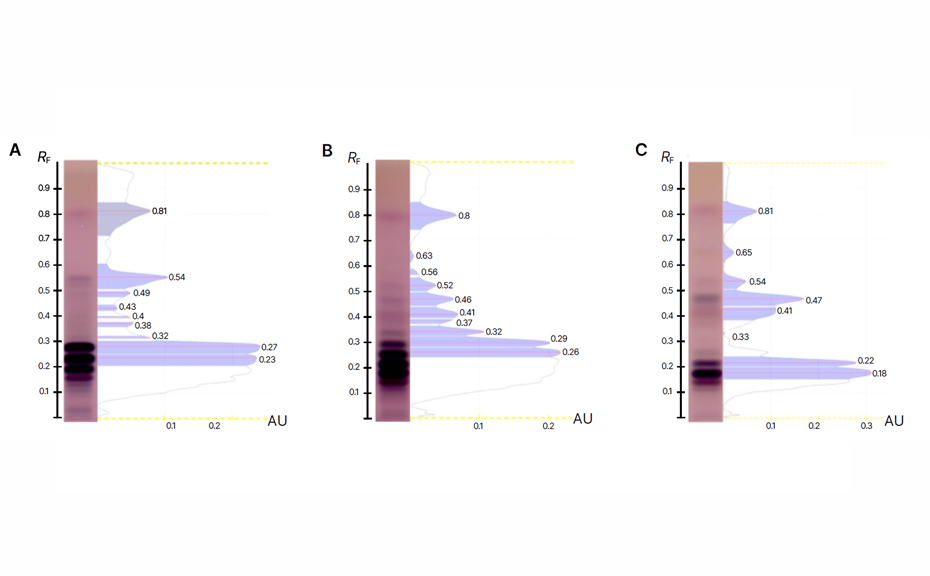
Fingerprints of tested oils with corresponding RF (represented with a red line), HPTLC plate in white light (transmission) after derivatization with anisaldehyde reagent; sunflower oil (A), rapeseed oil (B), and palm oil (C); (https://creativecommons.org/licenses/by/4.0/legalcode)
Fingerprints of tested oils with corresponding RF (represented with a red line), HPTLC plate in white light (transmission) after derivatization with anisaldehyde reagent; sunflower oil (A), rapeseed oil (B), and palm oil (C); (https://creativecommons.org/licenses/by/4.0/legalcode)
The following HPTLC chromatograms reveal the detection of adulteration in sunflower oil. Samples adulterated with cotton, safflower, corn, sesame, and soy oils were analyzed, and the corresponding RF values for each adulterant are marked with dashed lines. Adulteration was detected at RF values specific to each adulterant, such as RF 0.38 for cotton oil and RF 0.49 for sesame oil. The clear distinction between authentic and adulterated sunflower oil samples demonstrates the sensitivity of the HPTLC method, which successfully detected adulteration at levels as low as 5%.
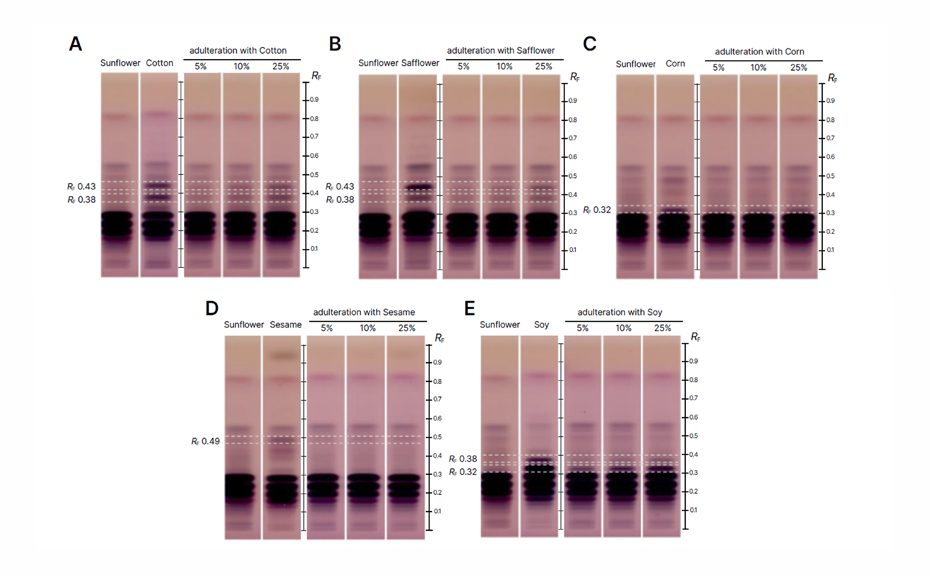
HPTLC chromatograms in white light (transmission) after derivatization with anisaldehyde reagent: Sunflower oil adulterated with cotton oil (A), safflower oil (B), corn oil (C), sesame oil (D), and soy oil (E) with the corresponding adulteration RF’s (represented with a dash lines); (https://creativecommons.org/licenses/by/4.0/legalcode)
HPTLC chromatograms in white light (transmission) after derivatization with anisaldehyde reagent: Sunflower oil adulterated with cotton oil (A), safflower oil (B), corn oil (C), sesame oil (D), and soy oil (E) with the corresponding adulteration RF’s (represented with a dash lines); (https://creativecommons.org/licenses/by/4.0/legalcode)
Adulteration was detected at RF values around 0.8 for mineral oil and paraffin wax, clearly distinguishing them from the authentic palm oil sample. The high sensitivity of the HPTLC method allowed for the detection of adulteration at levels below 5%, demonstrating its effectiveness in identifying hazardous non-edible oil contaminants such as mineral oils.
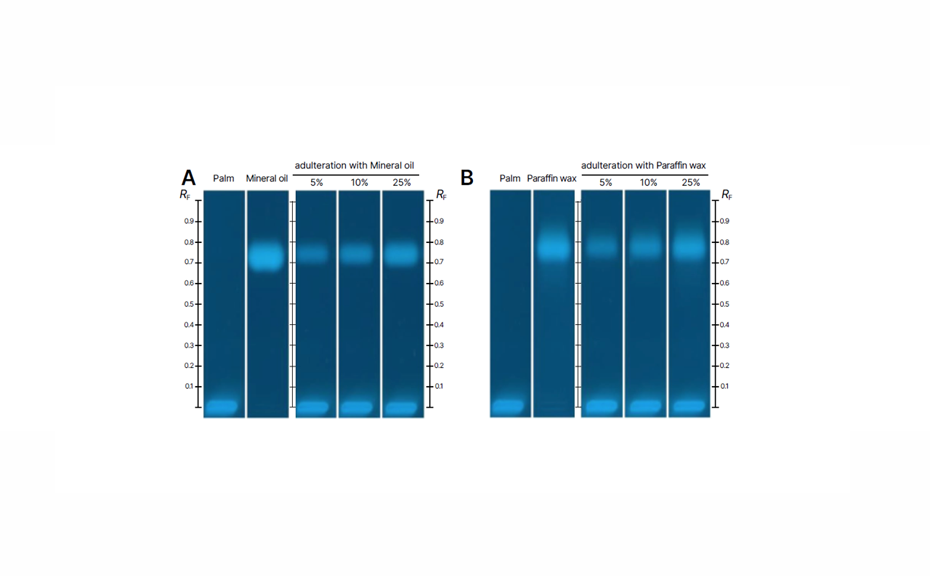
HPTLC chromatograms in UV 366 nm after derivatization with primuline reagent: Palm oil adulterated with mineral oil (A) and paraffin wax (B); (https://creativecommons.org/licenses/by/4.0/legalcode)
HPTLC chromatograms in UV 366 nm after derivatization with primuline reagent: Palm oil adulterated with mineral oil (A) and paraffin wax (B); (https://creativecommons.org/licenses/by/4.0/legalcode)
Adulteration was detected at RF values around 0.8 for mineral oil and paraffin wax, clearly distinguishing them from the authentic palm oil sample. The high sensitivity of the HPTLC method allowed for the detection of adulteration at levels below 5%, demonstrating its effectiveness in identifying hazardous non-edible oil contaminants such as mineral oils.
Conclusion
HPTLC proved to be a valuable tool for detecting adulteration in edible oils, offering a high-throughput, reliable, and relatively simple method. The method is well-suited for industrial applications, ensuring food quality and authenticity in the global edible oil market.
Literature
[1] Paul Rogeboz et al. Food Analytical Methods (2024) 17:1336–1347
Contact: Paul Rogeboz, Société des Produits Nestlé SA, Nestlé Research, 1000, Lausanne, Vers-Chez-Les-Blanc, Switzerland, paul.rogeboz@rd.nestle.com
To identify the propolis type, a simplified, rapid, low-cost, low-environmental impact, and easily adoptable analytical methodology was developed, extending to the standardization of selected neuroprotective components in Indian propolis. The versatility of HPTLC, with various derivatizing reagents and orthogonal detection capabilities, allows for increased applications. With the advent of thin-layer chromatography-effect directed analysis, it enables direct screening on the TLC plate, establishing preliminary evidence of the biological activities. Thus, this HPTLC method is valuable for rapid chemical profiling and simultaneous screening of antioxidant and anticholinesterase activities of Indian propolis. Also, educating beekeepers about its medicinal value can help them generate additional revenue.
HPTLC plates silica gel 60 F254 (Merck), 20 x 10 cm are used.
1.0-10.0 μL of standard solutions (7-point calibration) and 2.0 and 5.0 μL of sample solutions are applied as bands with the Linomat 5 (with N2). Plate layout: 15 tracks, band length 6.0 mm, distance from left plate edge 15.0 mm, track distance 11.4 mm, distance from the lower edge 8.0 mm.
Plates are developed in the twin-trough chamber with chamber saturation for 30 min (with filter paper) and development with toluene ‒ ethyl acetate ‒ formic acid 74:26:5 (V/V) to the migration distance of 80 mm (from the lower edge), followed by drying for 5 min.
Note: The derivatization was conducted on three different developed plates.
The developed plate is immersed into a 2,2-diphenyl-1-picryl hydrazyl solution (DPPH – 0.25 % (W/V) in methanol), stored in the dark for 30 min. The yellow zones captured against purple background are an indicator of antioxidant components when visualized in white light. The Ellman assay protocol was used wherein the developed plate is first immersed in a solution of 5,5′-dithiobis-2-nitrobenzoic acid (DTNB) and acetylthiocholine iodide (ATCI) (1 mM DTNB and 1 mM ATCI in buffer A) until the plate was saturated, dried for 5 min and then around 3-4 mL of acetylcholinesterase enzyme solution (Electrophorus electricus – AChE – 3 U/mL) is sprayed onto the plate. The white band on the plate is an indicator of acetylcholinesterase inhibition.
Images of the plate are captured with the TLC Visualizer 2 in UV 254 nm, UV 366 nm, and white light.
The HPTLC fingerprint image of the various propolis extracts is shown, and the profiles are key indicators of the diversity in vegetation across different regions. The sample coded HAR was mainly of ‘O-type’ propolis due to the presence of flavonoids like chrysin, galangin, pinocembrin, as well as non-flavonoids like p-coumaric acid, matching the characteristic bands of the standard when derivatized with various reagents. Interestingly, the applicability of the method on two marketed products presented a similar fingerprint to that of the HAR extract.
The optimized method is found to be precise (%RSD ≤ 2.0 %), accurate (90‒110 %), linear over the concentration ranges (r2 ≥ 0.995), sensitive and robust resulting in the RF values of 0.235, 0.353, 0.552, 0.606, and 0.655 for luteolin, p-coumaric acid, chrysin, galangin, and pinocembrin, respectively. Pinocembrin (2.30 ± 0.12 % W/W) and galangin (5.78 ± 0.30 % W/W) are found in the highest concentrations in the HAR sample. The m/z values of the molecular ion and fragment ions from the isolated sample bands matched those of the standards, further confirming the identity of the peaks. The bands with RF values corresponding to chrysin, galangin, and pinocembrin showed strong antioxidant activity, as indicated by bright yellow zones against a purple background, while the white bands in the extract fingerprint that appeared along the plate following the Ellman’s assay are indicative of acetylcholinesterase inhibitors.
Thus, the developed analytical method with orthogonal capabilities can be universally applied to different propolis extracts and formulated propolis products as a quick screening method for fingerprint and neuroprotective profiling.
SMOOTH & PRECISE OPERATION
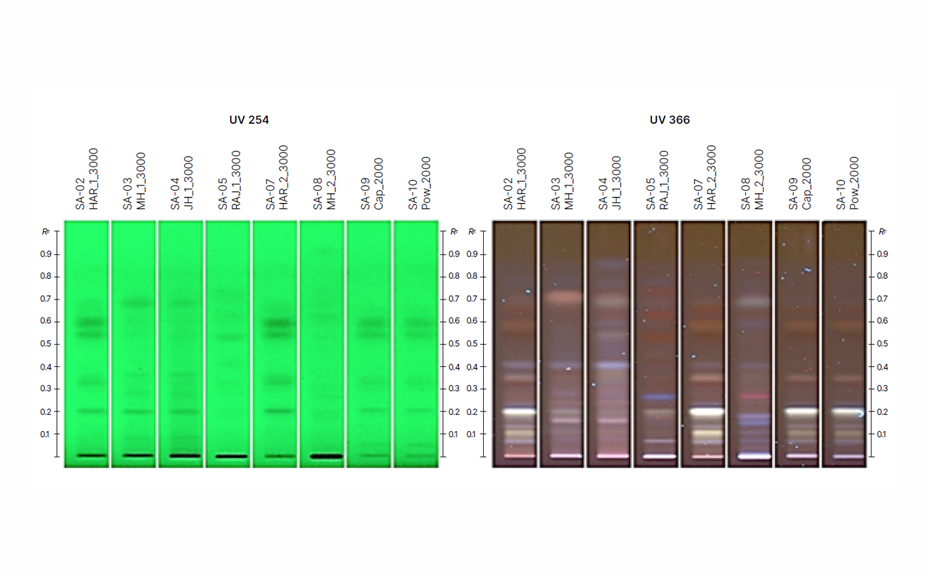
01
HPTLC fingerprint image of propolis extracts collected from different regions in India and marketed samples in UV 254 nm and in modified UV 366 nm before derivatization (enhanced contrast)

02
HPTLC fingerprints of HAR extracts pre- and post-derivatization in different illumination modes
[1] Sankaran, S. et al. (2024) J Planar Chromat 37 (3), 233–245
[2] Bankova, V. et al. (2019) J Apic Res 58, 1–49
[3] Sankaran, S. et al. (2023) J Biol Active Prod Nat 13, 76–93.
Further information is available in the article published “Sustainable instrumental thin-layer chromatography-based methodology for standardization of neuroprotective components in propolis collected from India” J Planar Chromat 37, 233–245 (2024). https://doi.org/10.1007/s00764-024-00307-x or on request from the authors.
Contact: Sandeep Sankaran, Department of Quality Assurance Techniques, Poona College of Pharmacy, Bharati Vidyapeeth (Deemed to be) University, Pune, Maharashtra 411038, India, sandeepsss1992@gmail.com
Food analytical methods, 2024
Authors: Paul Rogeboz, Hélia Latado, Ajay Sharma, Neha Chaubey, Shalu Kadian, Enrico Chavez, Thi Kieu Tiên Do, Mathieu Dubois, Francesca Giuffrida, Amaury Patin & Maricel Marin-Kuan
Chemical warfare agents present a considerable threat to human health, inducing a spectrum of symptoms ranging from irritation to fatality. It is imperative for law enforcement agencies and military personnel to possess the knowledge and tools required to detect and prevent exposure to these hazardous substances. There are various methods to categorize chemical warfare agents, one common approach is to categorize them based on the primary symptoms they cause. Nerve agents, for instance, are organic chemicals that disrupt the mechanisms through which nerves convey messages to organs. This disruption arises from the inhibition of acetylcholinesterase (AChE), an enzyme facilitating the breakdown of acetylcholine.
Blistering agents, also known as vesicants, are chemical warfare agents that induce skin blisters, eye damage, and respiratory harm. Typically, these agents manifest as oily liquids that can persist on surfaces for extended durations. Exposure to blistering agents can lead to severe burns, lung damage, and even death. In contrast, irritant agents elicit irritation on the skin, eyes, and respiratory system. Although less lethal than nerve agents and blistering agents, irritant agents can still inflict significant harm on exposed individuals. Examples of irritant agents include substances like chlorine gas, phosgene gas, and tear gas.
Arsenic agents represent another category of chemical warfare agents capable of causing substantial harm to human health. Exposure to arsenic agents can result in symptoms ranging from irritation to death.
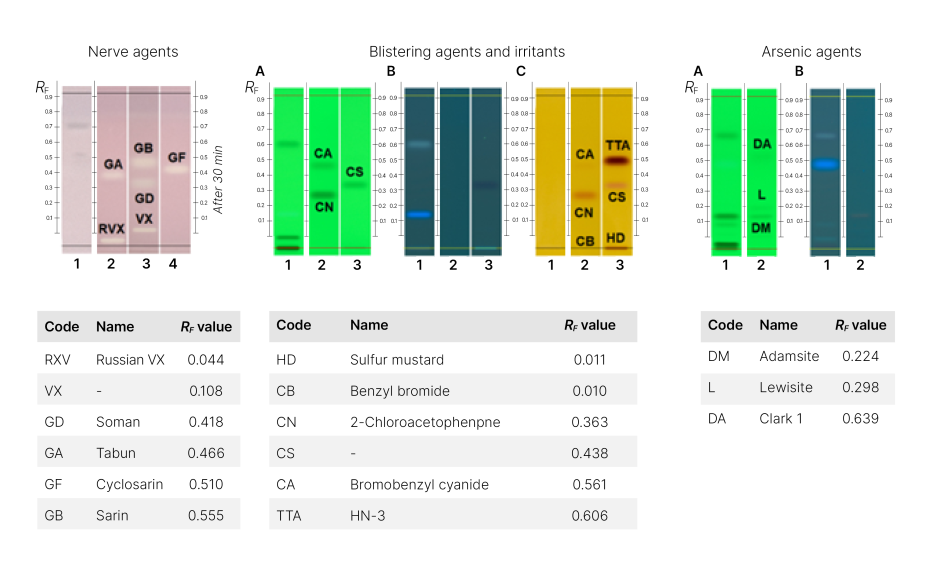
HPTLC is a reliable and widely used analytical technique for the identification of chemical warfare. HPTLC separates the individual components of a mixture, making it possible to identify specific nerve agents such as Russian VX (RVX), O-ethyl S-(2-diisopropylaminoethyl) methylphosphonothioate (VX), Soman (GD), Tabun (GA), cyclosarin (GF), and sarin (GB) based on their characteristic retention factor (RF) values [1]. TLC methods were transferred to HPTLC.
For six blistering agents and irritants, namely sulfur mustard (HD), HN-3 (TTA), 2-chlorobenzylidenemalononitrile (CS), 2-chloroacetophenone (CN), bromobenzyl cyanide (CA), and benzyl bromide (CB) [2], as well as three arsenic agents Lewisite (L), Clark 1 (DA), and Adamsite (DM) [3], their initial TLC methods were successfully transferred to HPTLC. This underscores the adaptability and efficacy of HPTLC in extending the capabilities of traditional TLC methods for the comprehensive analysis of chemical warfare agents.
Standard solutions
Individual standard solutions were prepared according to the table below, and for quantification purposes each solution was applied at different application volumes to generate a calibration curve.
System Suitability Test (SST): the ready-to-use solution of Universal HPTLC mix (UHM) was prepared in house according to [4] and applied on track 8 of each plate.

HPTLC plates silica gel 60 F254 (Supelco), 20 × 10 cm are used.
Samples are applied as bands with the Automatic TLC Sampler (ATS 4), 15 tracks, band length 8.0 mm, distance from left edge 20.0 mm, distance from lower edge 8.0 mm.
Plates were developed with the following three developing solvents in ADC 2 with activation of the plate at 33 % relative humidity for 10 min using a saturated solution of magnesium chloride. For nerve agents, acetone – cyclohexane – ethyl acetate – methanol 1:5:3:0.2 (V/V), for blistering agents and irritants, toluene, and for arsenic agents, cyclohexane – dichloromethane – methanol 7:2:1 (V/V), are used as developing solvents with 20 min chamber saturation (with saturation pad). The developing distance for all three methods was 70 mm (from the lower edge). Plates were dried for 5 min.
Post-chromatographic derivatization
For nerve agents:
1. Spraying solution A: Acetylcholinesterase
Reagent preparation:
Dissolve 55.0 mg of acetylcholinesterase (55 mg = 150 U) in 100.0 mL of buffer solution (dissolve 19.0 g of Na2HPO4 x 12 H2O and 1.8 g of KH2PO4 in 1.0 L of de-ionized water (pH approx. 7.4)).
Reagent use:
Spray the plate with 4.0 mL of spraying solution A with the Derivatizer, yellow nozzle, spraying level 4, and leave the plate (horizontal; outside of the Derivatizer) for 15 min at room temperature.
[Note]: with 4.0 mL, the plate should not dry out.
2. Spraying solution B: Fast blue salt
Reagent preparation:
Mix 40.0 mL of fast blue solution (100.0 mg of fast blue salt B in 40.0 mL of de-ionized water) with 10.0 mL of 1-naphthyl acetate solution (25.0 mg of 1-naphthylacetate in 10.0 mL of ethanol).
Reagent use:
Spray the plate with 2.0 mL of praying solution B with the Derivatizer, yellow nozzle, spraying level 4, and record the images after 30 min.
For blistering agents and irritants (optional):
1. Spraying solution C: 4-(4’-Nitroenzyl)-pyridine solution
Reagent preparation:
Dissolve 5.0 g of 4-(4’-nitrobenzyl)-pyridine in 100.0 mL of ethanol.
2. Spraying solution D: Benzofurazan-(1)-oxide solution
Reagent preparation:
Dissolve 1.0 g of benzofurazan-(1)-oxide in 100.0 mL of ethanol.
Reagent use:
Spray the plate with 2.0 mL of spraying solution B with the Derivatizer, yellow nozzle, spraying level 4, and record the images after 30 min.
3. Spraying solution E: NaOH solution
Reagent preparation:
Dissolve 4.0 g of NaOH in a mixture of 50.0 mL of de-ionized water and 50.0 mL of methanol.
Reagent use:
Spray the plate with spraying solution C with the Derivatizer (yellow nozzle, 3.0 mL, spraying level 4), heat at 150 °C for 30 s, and immediately record images. Spray the plate with spraying solution D with the Derivatizer (yellow nozzle, 3.0 mL, spraying level 3), and then with spraying solution E with the Derivatizer (yellow nozzle, 3.0 mL, spraying level 6), and record the images within the next 2 min.
Documentation
TLC Visualizer in UV 254 nm, UV 366 nm, and white light prior to derivatization, and UV 366 nm, and white light after derivatization (as needed).
Densitometry
For the UHM, TLC Scanner 4 and visionCATS, absorbance measurement at 254 nm, slit dimension 5.00 mm x 0.20 mm, scanning speed 50 mm/s, and in fluorescence mode at 366>/400 nm. For the other substances, each standard is detected at their maximum of absorption as described in the following table.
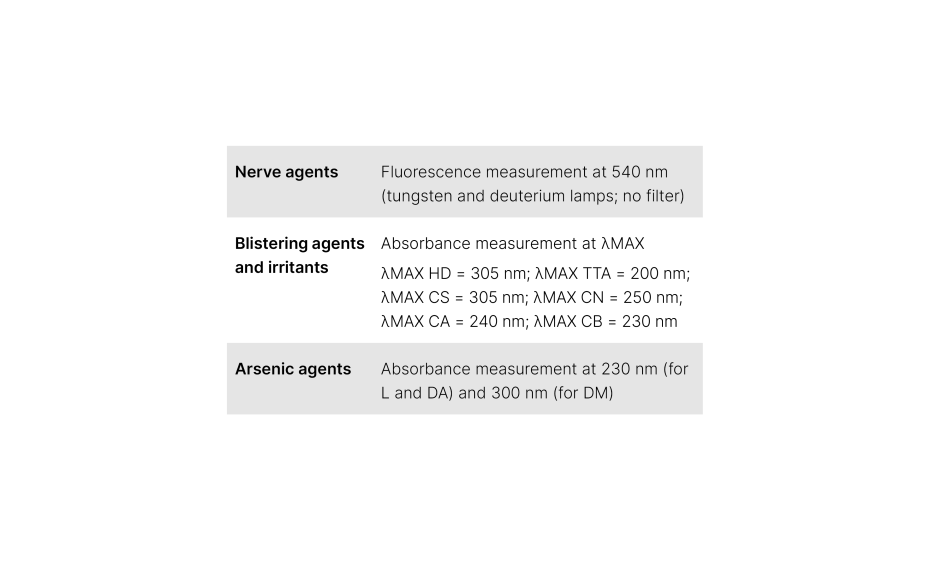
For each method, the UHM was used as SST and the RF values to obtain for each method are described as follows:
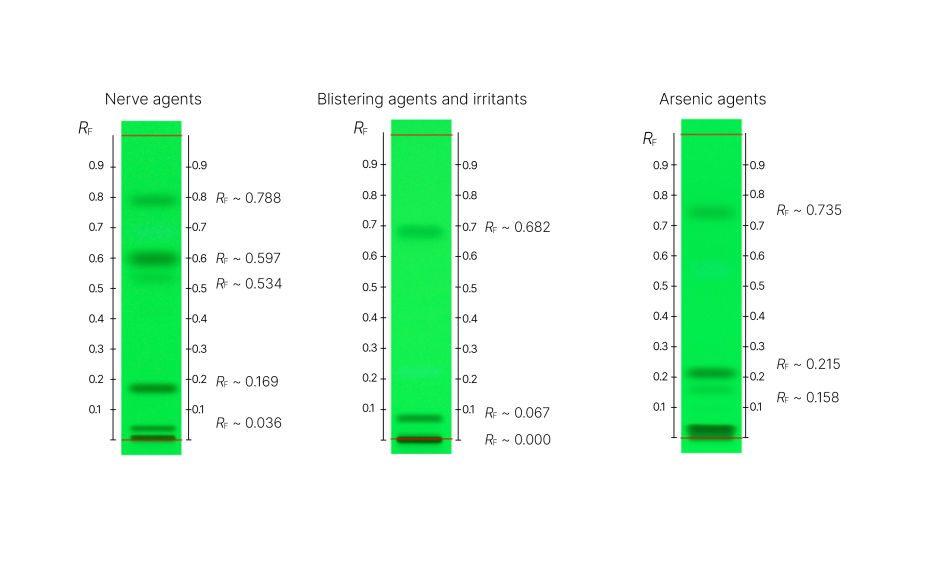
01
RF values to obtain for SST using the UHM for each method
02
HPTLC chromatograms and RF values for each nerve agent, blistering agents and irritants, and arsenic agents
For nerve agents, a large-scale untargeted screening of samples was developed, involving the detection of toxic substances without specific identification
In this approach, each sample (utilizing reference substances in our example) was applied at different Y positions, forming a zone equivalent to a 1.0 mm band, with varying application volumes. In our example, the screening was applied on a 20 x 10 cm plate, but the screening could also be applied to a 20 x 20 cm plate.
Following the application, no development was conducted, but the entire plate underwent derivatization. Positive zones were observed as yellow against a pink/violet background. This test revealed that each nerve agent was still detectable at very low absolute quantities (amount on the plate):
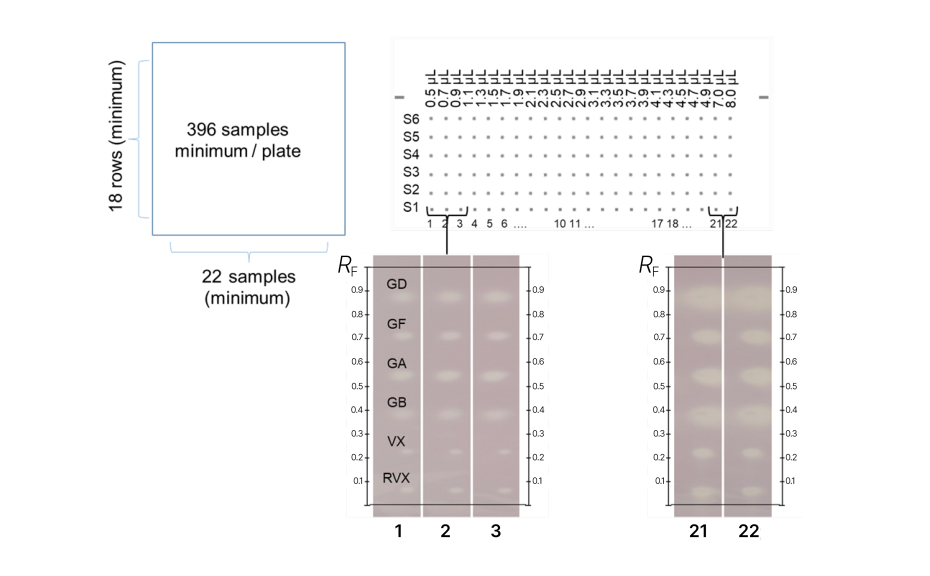
Protocol developed for large-scale untargeted screening of samples for detection of nerve agents (top), and example with reference substances in white light after derivatization (bottom). RVX (0.5 ng/μL) was applied at Y = 10 mm, VX (0.5 ng/μL) at Y = 20 mm, GB (0.25 ng/μL) at Y = 30 mm, GA (0.5 ng/μL) at Y = 40 mm, GF (0.02 ng/μL) at Y = 50 mm, and GD (0.05 ng/μL) at Y = 60 mm
Conclusion
The examples above show that HPTLC is a valuable tool for identifying nerve agents, blistering agents and irritants, as well as arsenic agents which are important for law enforcement and military personnel in preventing chemical warfare. HPTLC’s format preserves the separated zones, allowing for further investigation including bioassays like acetylcholinesterase inhibition. Additionally, the use of HPTLC instruments reduces the need for analysts to physically interact with toxic samples, enhancing safety.
Literature
[1] CAMAG Application note A-142.1: Identification and quantification of arsenics agents L, DA and DM by HPTLC.
[2] CAMAG Application note A-143.1: Identification and quantification of blistering agents and irritants HD, TTA, CS, CN, CA and CB by HPTLC.
[3] CAMAG Application note A-144.1: Identification and quantification of nerve agents RVX, VX, GD, GA, GF and GB by HPTLC, and methodology for a large-scale untargeted screening.
[4] T. K. T. Do et al., J Chromatogr A (2021) 1638
Contact: Dr. Tiên Do, Sonnenmattstrasse 11, 4132 Muttenz, Switzerland, tien.do@camag.com
HPTLC is a straightforward analytical technique that offers numerous advantages. While the technique follows the same concept of separating mixture components between two phases (mobile phase and stationary phase), it differs from other liquid chromatographic techniques in the fact that a gas phase is present during and, indeed, influences the development process [1]. This property has always posed a challenging issue for controlling the outcome of the separation. Moreover, the fact that a broad spectrum of solvents can be used means gas phase control holds great promise for resolving complex matrix separations.
In order to investigate this, the Module DEVELOPMENT (a component of the CAMAG® HPTLC PRO System [2]) was employed in this study. The Module not only allows the generation and introduction of a gas phase of varying composition into the development chamber but also provides control over the timing and power settings of the pump used to build up the gas phase. The Module is equipped with three separate solvent bottles that enable the generation of gas phase from either the same solvents used for plate development or from different solvents. Additionally, the Module can be configured to introduce the generated gas phase at two distinct stages, prior to the start of the development (referred to as pre-conditioning) and/or during the development process (referred to as conditioning). These features provide useful tools to control the gas phase throughout the development process.
This study aims to investigate whether it is possible to manipulate the gas phase to attain a desired chromatographic separation. To achieve this objective, we sought to control the gas phase in a way that we can obtain RF values based on the Universal HPTLC mix (UHM), a mixture of chemicals for system suitability testing, that are comparable to (ΔRF ≤ 0.05) those previously measured using the ADC 2 [3].
Standard solutions
The ready to use solution of UHM was prepared in house according to [4] and applied on track 8 (middle track) of each plate.
Chromatogram layer
HPTLC plates silica gel 60 F254 (Supelco), 20 × 10 cm are used.
Sample application
2.0 µL of UHM solution are applied as bands with the HPTLC PRO Module APPLICATION, 15 tracks, band length 8.0 mm, distance from the left edge 20.0 mm, track distance 11.4 mm, distance from the lower edge 8.0 mm. The default settings of methanol as sample solvent are used. The first rinsing step (bottle 1 solvent) is done with methanol – acetonitrile – iso-propanol – water – formic acid 250:250:250:250:1 (V/V) and the second rinsing step (bottle 2 solvent) is done with methanol – water 7:3 (V/V).
Chromatography
Images of the plates are captured with the TLC Visualizer in UV 254 nm.
Documentation
Results and discussion
It is known that the gas phase surrounding the HPTLC plate during the development process can significantly influence the chromatographic separation. The HPTLC PRO Module DEVELOPMENT has a unique chamber design compared to the chambers used in the ADC 2 or for manual development. These differences can lead to changes in the rate of evaporation and the concentration of the developing solvent, which may result in differences in the pattern of separations. Therefore, it may be expected that the RF values measured using the HPTLC PRO Module DEVELOPMENT will exhibit some deviations from those obtained through ADC 2 or manual development.
To explore the effects of the gas phase on compound separations, our goal in this study was to achieve RF values similar to those obtained using the ADC 2 method.
Key aspects of the study involve:
An initial experiment (HPTLC PRO M1) was conducted without using pre-conditioning or conditioning. In comparison to the ADC 2 results, the overall RF values were different. However, compounds [e], [f], and [h] exhibited average RF values within the specified control limits. Notably, ΔRF was higher for compound [g] (~ 0.06).
These findings suggest that the migration pattern for all compounds does not behave uniformly. The development without the use of the gas phase leads to increased RF values for compounds in the lower half of the plate, and to decreased RF values for compounds in the upper half of the plate.
The challenge now is to control the retention of each of the four compounds individually on the plate solely based on gas phase control.
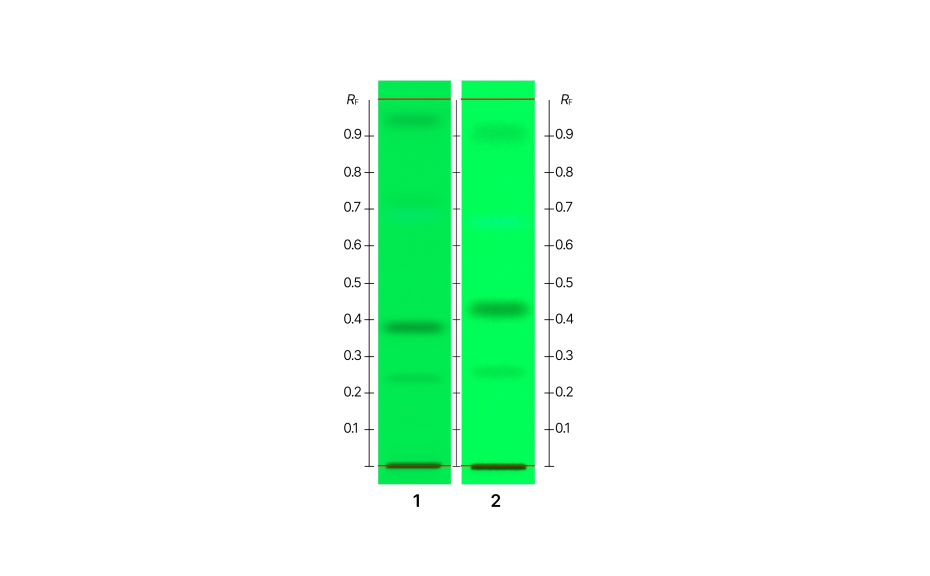
HPTLC chromatogram of UHM after development. 1: Experiment ADCRF 2, 2: Experiment HPTLC PRO M1
Based on this initial information, three methods were developed to evaluate the effect on the ΔRF.
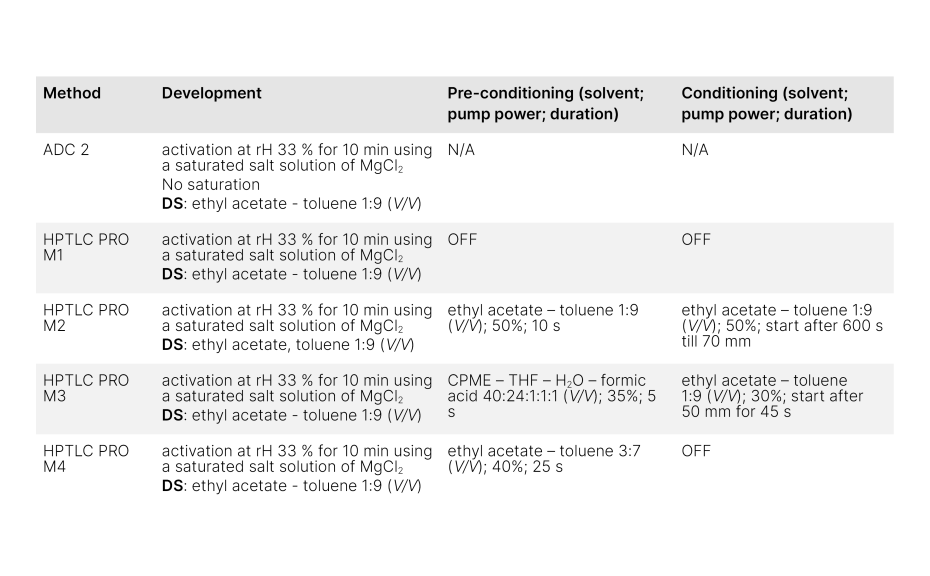
Methods used to study active gas phase control
The method HPTLC PRO M2 focused on using the same solvent for both, the developing solvent and for gas phase generation. Initially, conditioning with the developing solvent was employed. However, the experiment revealed that initiating conditioning at various migration distances (while keeping the pump power constant) significantly affected the RF values of individual substances.
For example, beginning conditioning at either 0 or 30 mm resulted in a substantial reduction of the RF value for zones located in the upper part of the plate, while starting conditioning at 50 mm exhibited less impact on these zones. Consequently, we decided to initiate conditioning after 50 mm, leading to an improvement of the RF values for most zones, except for compound [f], which required the use of a pre-conditioning step. Previous studies have shown that conditioning in normal phase HPTLC usually increases the RF values and pre-conditioning lowers them.
Ultimately, increasing the pre-conditioning duration from 10 to 30 s corrected the RF value for compound [f], but this came at the expense of reduced RF values for compounds [g] and [h].
Those data highlight the various parameters that can be used to regulate the gas phase. It also reveals that substances respond differently to each given experimental condition, indicating that the chemical properties of the compounds play a role in regulating the gas phase.
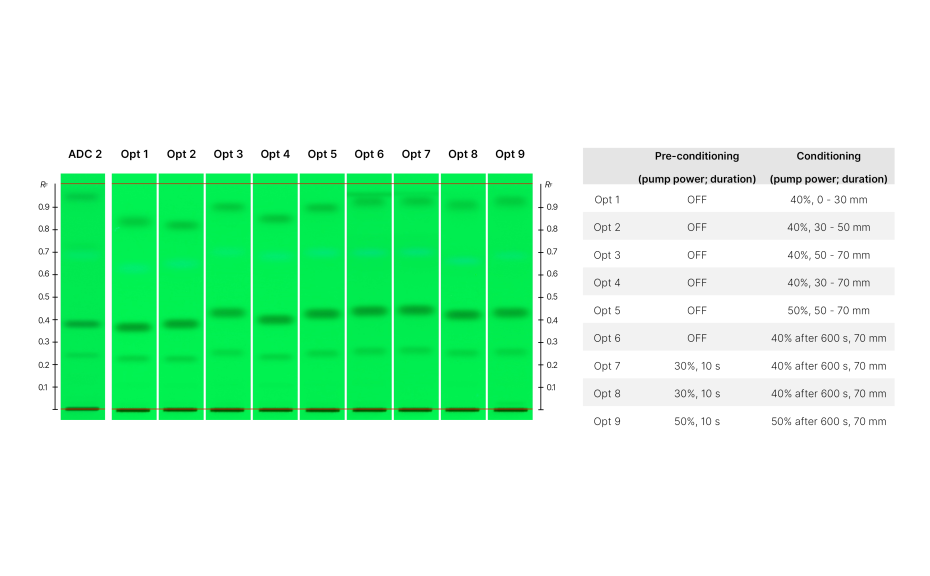
HPTLC chromatograms show results obtained at different conditions with method HPTLC PRO M2
Similar optimization processes were employed in the other two approaches (optimization data not shown). However, in these two approaches we demonstrated how to control the gas phase with solvents that are different from the developing solvent. One approach involved entirely different solvents, adopted from [3] (referred to as HPTLC PRO M3), while the other maintained the same composition but different solvent proportions (referred to as HPTLC PRO M4).
Notably, the fourth approach (HPTLC PRO M4), which uses ethyl acetate – toluene 3:7 (V/V) for pre-conditioning, yielded the most favorable outcome. In this approach, no conditioning is required and in contrary to the common tendency for pre-conditioning to decrease RF values (due to the known building of virtual fronts), our study revealed an anomalous outcome where RF values for compounds other than [g] experienced an increase in RF value. By exploring these alternative solvent combinations, we can expand our understanding about the effect of the gas phase composition and its subsequent impact on chromatographic performance.
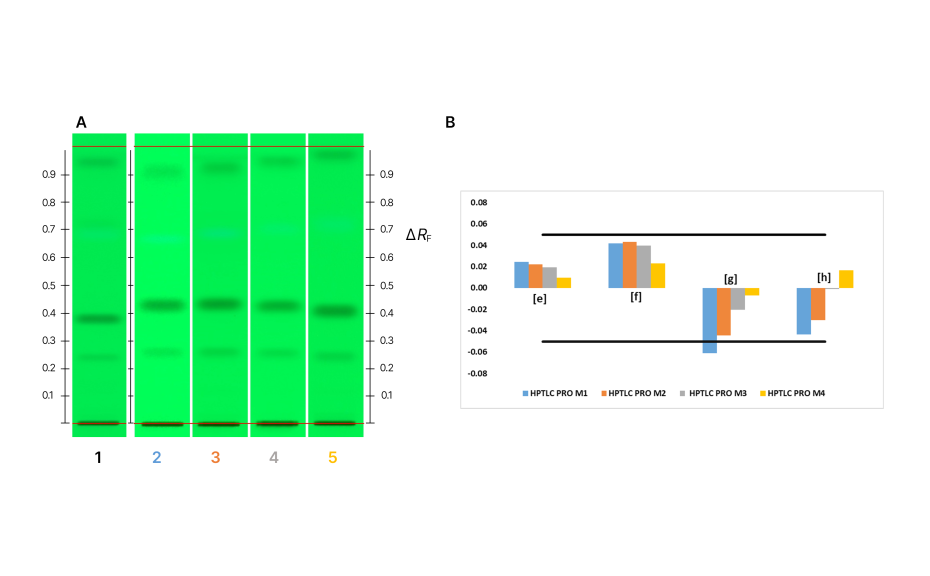
HPTLC chromatograms of the UHM after development with different conditions (A): track 1: ADC 2 (standard conditions), track 2: HPTLC PRO M1, track 3: HPTLC PRO M2, track 4: HPTLC PRO M3, track 5: HPTLC PRO M4; Control chart for ΔRF (B)
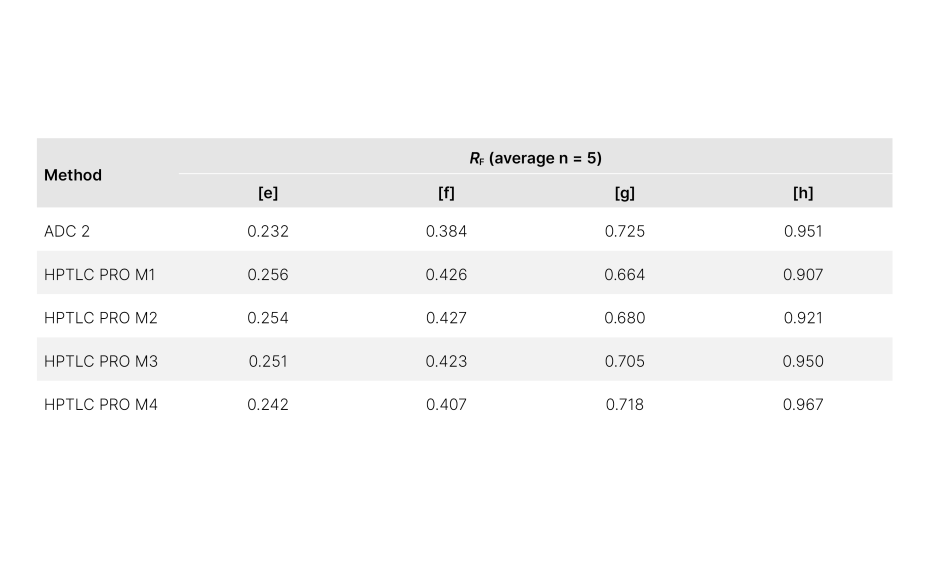
RF values obtained from methods conducted in this study
Conclusion
This study emphasizes the essential role of the gas phase in regulating the development process and extends its significance beyond the establishment of standardized chromatographic procedures for HPTLC analysis.
Furthermore, this study shows, that it is possible to control the gas phase. By optimizing the composition of the gas phase, the pump power used to build up the gas phase, and the duration of the gas phase using the HPTLC PRO Module DEVELOPMENT, we demonstrated how the control of the gas phase allows the customization of the retention of each of the target compounds in specific regions of the chromatogram. This results in the achievement of the desired separation pattern through three distinct approaches.
This groundbreaking work highlights the critical role of the gas phase in controlling the development process, introducing new possibilities for strengthening and enhancing the selectivity of the gas phase on the development. These concepts, previously not fully explored, represent a significant step towards a deeper understanding of the complexities involved in pre-conditioning and conditioning processes within Thin-Layer Chromatography systems.
Notably, the fourth approach (HPTLC PRO M4), which uses ethyl acetate – toluene 3:7 (V/V) for pre-conditioning, yielded the most favorable outcome. In this approach, no conditioning is required and in contrary to the common tendency for pre-conditioning to decrease RF values (due to the known building of virtual fronts), our study revealed an anomalous outcome where RF values for compounds other than [g] experienced an increase in RF value. By exploring these alternative solvent combinations, we can expand our understanding about the effect of the gas phase composition and its subsequent impact on chromatographic performance.
Literature
[1] E. Reich et al., High-Performance Thin-Layer Chromatography for the Analysis of Medicinal Plants (2007).
[2] CAMAG CBS 123. Introducing CAMAG HPTLC PRO.
[3] T. K. T. Do et al. J. Planar Chromatogr. – Mod. TLC (2022) 299
[4] T. K. T. Do et al., J Chromatogr A (2021) 1638
Contact: Dr. Ehab Mahran, CAMAG, Sonnenmattstrasse 11, 4132 Muttenz, Switzerland, ehab.mahran@camag.com
Mr. Ramakant Yadav, an application specialist under the guidance of Akshay Charegaonkar (Managing Director), works at Anchrom Enterprises Pvt Ltd, Mumbai, India. The company specializes in instrumental Planar Chromatography and is renowned for its expertise in developing novel, quantitative, and regulatory-compliant analytical methods for a wide range of products, including pharmaceutical formulations, APIs, herbal products, food items, organic intermediates, and dyes. Mr. Yadav finds HPTLC advantageous due to its rapidity, ease of use, cost-effectiveness, and data outputs such as plate images both pre- and post-chromatographic derivatization, along with the ability to evaluate data through image, profile, and spectrum comparisons.
Crocus sativus L. commonly known as saffron, is a perennial stemless herb that is widely cultivated in Iran, India and Greece. It is obtained by drying the stigma of C. sativus L., which belongs to the Iridaceae family. Saffron plays a pivotal role in modern and traditional medicine, it is utilized for the prevention and treatment of various diseases and has anti-hypertensive, antioxidant, antidepressant, and anti-inflammatory activity. This precious spice holds a broad spectrum of applications in the food and cosmetic industries, serving as both a flavoring and coloring agent. The quality of saffron is affected by various factors, such as cultivation regions, climate, drying process, and storage conditions. However, the high value of this product makes it very susceptible to economic adulteration, which involves the mixing of low-quality spices with saffron, the addition of plant materials, and the use of natural or artificial colorants to imitate the color of saffron.
HPTLC is widely implemented in the food industry as a convenient and low-cost approach for separations of different chemical components, such as adulterants and contaminants. It is well suited for adulteration studies, because it is inexpensive and time-saving. By HPTLC, 15-20 samples can be detected simultaneously on one plate in about 20-30 minutes. The solvent consumption is only about 20 mL for those 20 samples and little waste is produced. Hence a method was developed for comparison of marketed saffron samples with pure saffron sample (BRM) to find possible adulterants.
Standard solutions
100 mg of Crocus sativus L. (Saffron BRM) is dissolved in 10 mL of 70% ethanol.
Sample preparation
100 mg of Crocus sativus L. (Saffron) marketed formulations are dissolved in 10 mL of 70% ethanol. The samples are vortexed thoroughly and centrifuged at 3000 rpm for 5 min. After centrifugation, the supernatant was collected and used for the application.
Chromatogram layer
HPTLC plates silica gel 60 F254 (Merck), 20 x 10 cm are used.
Sample application
2.0 and 5.0 µL of sample solutions and 2.0 and 5.0 µL of standard solutions are applied as bands with the Automatic TLC Sampler (ATS 4), 15 tracks, band length 8.0 mm, distance from the left edge 20.0 mm, track distance 11.4 mm, distance from the lower edge 8.0 mm.
Chromatography
Plates are developed in the ADC 2 with chamber saturation (with filter paper) for 20 min and after activation at 33% relative humidity for 10 min using a saturated solution of magnesium chloride, development with ethyl acetate – methanol – water 18:4:3 (V/V) to the migration distance of 70 mm (from the lower edge), followed by drying for 5 min.
Post-chromatographic derivatization
The plate is pre-heated at 105 °C for 3 min using the TLC Plate Heater and is then sprayed with 3 mL of natural product A reagent (1 g of 2-aminoetheyl diphenylborinate in 200 mL of ethyl acetate) using the Derivatizer.
Documentation
Images of the plate are captured with the TLC Visualizer in UV 254 nm, UV 366 nm, and white light.
The analysis conducted on saffron samples involved a comparison between saffron (BRM) and saffron available in the market (branded and non-branded). Upon developing the chromatographic plate, it was observed that the fingerprints of all the saffron samples from the market were identical to that of the saffron (BRM). Notably, no adulterants were detected in any of the saffron samples, as there were no discernible colored bands observed apart from the characteristic fingerprint pattern.
Further investigation involved taking spectra of the major bands detected in all the saffron samples for the purpose of comparison. The results of this spectral analysis revealed that all the spectra from the different saffron samples, including both BRM and market-sourced saffron, matched identically. This suggests consistency and purity among the saffron samples, reinforcing their authenticity and quality.
01
HPTLC fingerprints: White light (A), UV 254 nm (B) & UV 366 nm (C) prior to derivatization, and white light after derivatization (D) and UV 366 nm after derivatization (E); Tracks 1 & 7: Saffron (BRM), tracks 2 & 8: marketed branded sample 1, tracks 3 & 9: marketed branded sample 2, tracks 4 & 10: marketed branded sample 3, tracks 5 & 11: marketed non-branded sample 1, and tracks 6 & 12: marketed non-branded sample 2
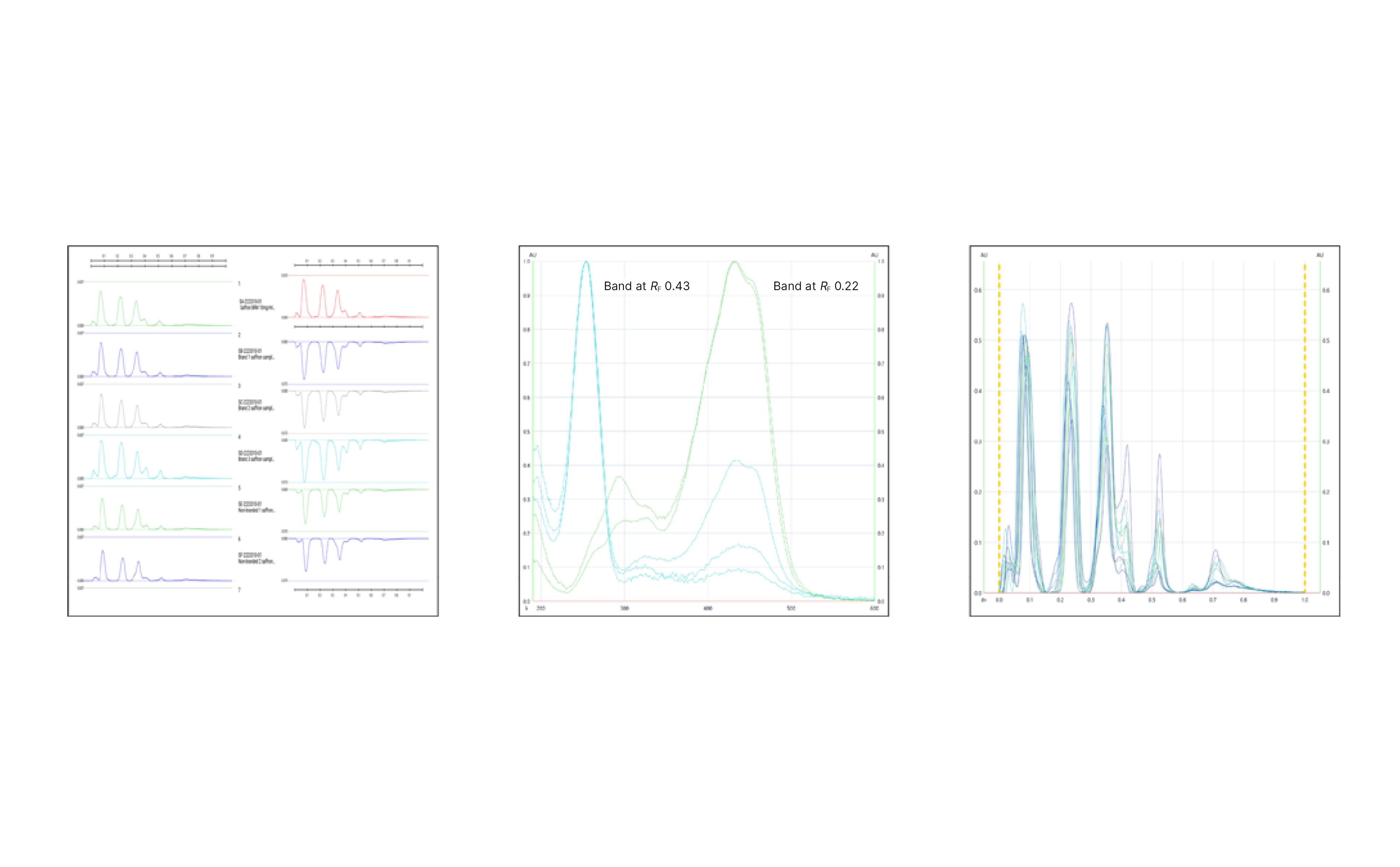
02
Left: Stack and flip view of saffron BRM and marketed saffron samples; middle: spectrum comparison of the two prominent zones found at RF 0.22 and 0.43 in both the marketed saffron samples and the BRM; right: HPTLC peak profiles for saffron BRM and marketed saffron samples
Literature
[1] American Herbal Pharmacopoeia
Contact: Mr. Akshay Charegaonkar, A-101, Shree Aniket Apartments, Navghar Road, Mulund East, Mumbai, Maharashtra 400081, India, hptlc[at]anchrom.in, www.anchrom.in
The bachelor thesis described below was conducted at the Department of Analytical Research / Development at WALA Heilmittel GmbH in cooperation with Esslingen University of Applied Sciences, Germany.
Dr.-Ing. Margit Müller leads the team of Analytical Development Intermediate and Finished Products at WALA Heilmittel GmbH (Bad Boll, Germany). She and her team are responsible for analytical methods and specifications for medicinal products and plant-based active pharmaceutical ingredients including herbal drugs. HPTLC and TLC methods are widely used for identity testing in quality control, including stability testing of WALA products.
Prof. Dr. Constanze Stiefel teaches instrumental analysis at the Faculty of Science, Energy and Building Services at Esslingen University of Applied Sciences. Her research focuses on the application and development of chromatographic methods, including HPTLC and effect-directed analysis to determine bioactive compounds, contaminants and residuals in food and cosmetics.
Alina Kaya studied Chemical Engineering / Color and Coatings at Esslingen University of Applied Sciences. The shown study represents her bachelor thesis, concluding her Bachelor of Science degree.
The presented study examines the implementation of software-based Design of Experiments (DoE) for robustness studies and method evaluation using the European Pharmacopoeia HPTLC method for identity testing of Arnica flower (monograph 1391, Ph. Eur. 11.0) as an example. DoE is expected to reduce practical effort in comparison to the traditional “one-factor-at-a-time” approach of experimentation. A statistically-based DoE approach aims at identifying significant factors and their interactions in relation to one or more response variables. Furthermore, it can be used to assess the robustness of the method against various factors or factor ranges.
The practical implementation of robustness studies is time-consuming. The use of DoE is intended to make these investigations more efficient by reducing practical effort while at the same time broadening method knowledge. Once a robust and valid parameter range has been identified by DoE, future method changes can be assessed with more confidence. The regulatory relevance of such variations can thus be reduced, which is advantageous because the testing of medicinal products must adhere either precisely to European Pharmacopoeia methods or require validation for every modified method. The use of DoE enables significant time and cost savings, making it highly interesting for pharmaceutical manufacturers.
Design of Experiments
The set-up of DoE-based experimental plans was carried out with the help of the software “Design Expert®” (version 22, StatEase, USA). Screening, characterization, and optimization studies were carried out.
A two-level factorial design was used for screening and characterization studies, investigating linear relations. Response surface methods were used for carrying out optimization studies, investigating more complex relationships such as quadratic relations. To take a closer look at the influence of the developing solvent components, a Mixture Design was used.
Standard solutions
Reference solution a: 1.0 mL of caffeic acid solution (1.0 mg/mL) and 1.25 mL of rutoside rihydrate solution (1.0 mg/mL) are diluted in methanol R to 5.0 mL.
Reference solution b: 500 µL of caffeic acid solution (1.0 mg/mL) and 625 µL of rutoside-trihydrate solution (1.0 mg/mL) are diluted in methanol R to 10.0 mL.
Reference solution c: 1.0 mL of chlorogenic acid solution (1.0 mg/mL) and 2.5 mL of hyperoside solution (1.0 mg/mL) are diluted in methanol R to 10.0 mL (used as system suitability test (SST)).
Sample preparation
2.00 g of powdered herbal drug (710) are extracted with 10.0 mL of methanol R, ultrasonically treated for 15 min, and filtered. The filtrate is used.
Chromatogram layer
HPTLC glass plates silica gel 60 F254 (Merck), 20 × 10 cm (cut into 10 × 10 cm) resp. 10 × 10 cm plates are used.
Sample application
2.0 µL of sample and reference solutions are applied as bands with Automatic TLC Sampler (ATS 4), band length 8.0 mm, distance from left edge 20.0 mm, track distance 12.0 mm, distance from the lower edge 8.0 mm.
Chromatography
Plates are developed up to 70 mm (from the lower edge) in a saturated 10 × 10 cm twin trough chamber with formic acid R – water R – ethyl acetate R 6:9:90 (V/V), followed by drying for 5 min with a cold air dryer.
Post-chromatographic derivatization
Plates are derivatized using the Derivatizer. After heating the plate at 105 °C for 5 min, the plate is sprayed while still warm, with 2.0 mL of diphenylboryloxyethylamine in methanol R (10 g/L) and 2.0 mL of Macrogol 400 R in methanol R (50 g/L), blue nozzle, praying level 3. The plate is air dried for 5 min.
Documentation
Images of the plate are captured with the TLC Visualizer in UV 366 nm after derivatization.
Results and discussion
In the first step of the investigation, critical risk factors were identified through a two-level factorial screening DOE design. As a result of the risk analysis, nine factors were identified as potentially critical for this HPTLC method. High level (+ 1) and low level (− 1) values of each risk factor were defined. The resolution between the two SST-components was selected as the response.
Before starting the experiments, a statistical power analysis was carried out using the software to ensure that the experimental plan had good predictive power. The power of a system should always be above 80%, which was confirmed in this case. Experimental runs were performed in the laboratory, and response values were measured.The measured responses were entered into the Design Expert software against respective experimental runs and data analysis was performed using half normal plots and Pareto charts.
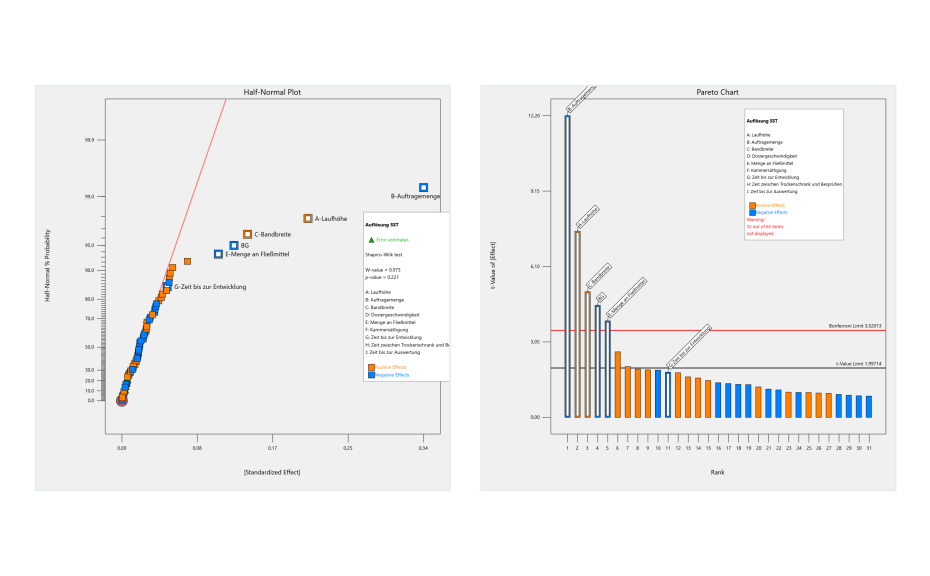
Half normal plot (left) and pareto chart (right) of two-level factorial screening design. Factors deviating from the red line of the half normal plot or the bars above the red line of the Pareto chart are statistically significant.
From the screening design, it was concluded that the application rate, development distance, volume of developing solvent, band length and the two-factor interaction between application rate and time until development are statistically significant. These identified risk factors need to be optimized to minimize their risk. Optimization was performed by DoE based on a central composite design. The central composite design was selected as response surface methodology to establish the relationship between the identified critical risk factors and resolution of the SST. Resolution values were added into the software against their respective experimental runs, and response surface analysis was performed with the help of variance analysis (ANOVA) and contour plots.
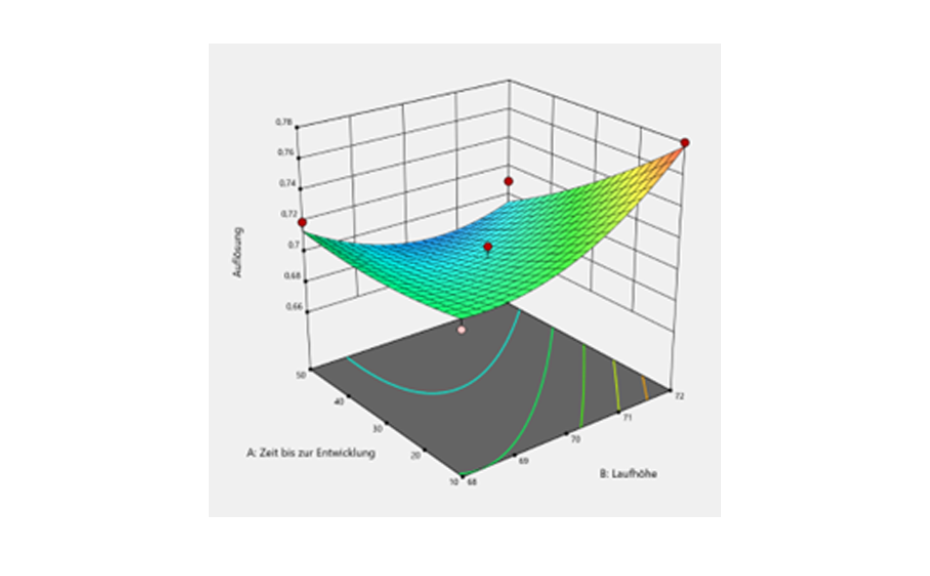
3D-contour plot of the experimental space
Although the optimization of significant factors was not successful in the context of this HPTLC-method, as no meaningful results could be generated, it can be said that the method is robust in the examined area because the resolution values of the different experiments changed only slightly.
For additional insights, an optimal mixture design study was carried out by investigating the robustness of the method with respect to formic acid and ethyl acetate concentration in the developing solvent. In addition to resolution of the SST components, the position of chlorogenic acid (substance of the SST) in the densitogram (expressed in mm) was selected as response parameter. With increasing amounts of ethyl acetate and decreasing amounts of formic acid, the resolution and the absolute position of chlorogenic acid decreased.
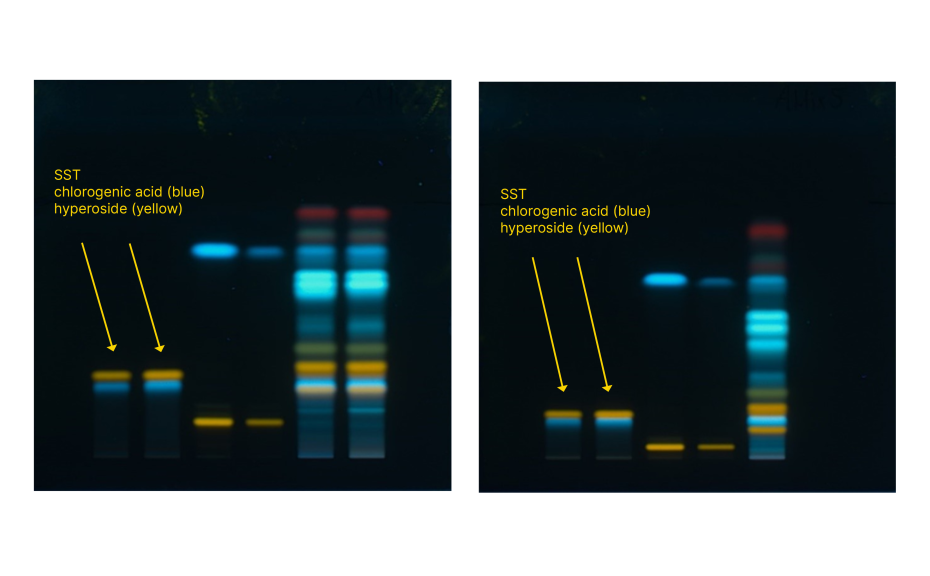
Comparison of SST resolution with the highest proportion of formic acid (left) and the lowest proportion of formic acid (right)
The application of DoE enables the efficient solution of different chromatographic problems in the field of TLC and HPTLC, in robustness studies of various method parameters, and in method validations, by creating systematic, precise experimental plans. The influence of various factors on any number of target variables can be investigated and evaluated in an experimental design. However, it must be noted that the number of tests increases with the number of factors to be investigated. The study showed that it was straightforward to assess the robustness of the selected TLC method across various factors. It was also possible to estimate the impact of method changes on the target variable within the area investigated by DoE.
Optimizing significant factors and minimizing their risks by determining a robust range was not possible for the presented method. However, this limitation may not apply to other methods. Various software programs offer a wide range of experimental design options. These include screening designs, response surface designs, mixture designs and many more. The generated data can be analyzed using a variety of statistical methods and analysis tools.
The results can be visualized through diagrams, graphs, and three-dimensional models. The results are statistically validated by the implementation of DoE-based experimental plans. However, it is essential to carefully consider the design of the experiment and the choice of factors and responses. The experimental conditions should be carefully controlled, and it should be ensured that the results are statistically valid and relevant to the intended field of application.
As a result, the thesis proved that by using a DoE-based approach the relevance of various robustness parameters and changes regarding developing solvent components can be assessed systematically and efficiently relating to the selected response parameter resolution of SST-components.
Literature
[1] Anderson, M. J. and Whitcomb, P. J. DOE simplified. Practical tools for effective experimentation. CRC Press. (2015)
[2] Anderson, M. J. RSM Simplified. Optimizing processes using response surface methods for design of experiments, second edition. CRC Press. (2017)
[3] Spangenberg, B. Quantitative Dünnschichtchromatographie. Eine Anleitung für Praktiker. Springer Spektrum (2014)
Contact:
Dr.-Ing. Margit Müller, WALA Heilmittel GmbH, Badwasen 2(T1), 73087 Bad Boll, Germany, margit.mueller[at]wala.de
Prof. Dr. Constanze Stiefel, Hochschule Esslingen, Kanalstraße 33, 73728 Esslingen, Germany, constanze.stiefel[at]hs-esslingen.de
Alina Kaya, alinameryemkaya[at]gmail.com