Simultaneous separation of fourteen antimalarial active pharmaceutical ingredients and three related impurities by HPTLC

Tobias is the HPTLC Product Manager at CAMAG and is responsible for the global support of instrumentation and workflows. Tiên is the Chief Scientific Officer at CAMAG, leading the company’s scientific activities.
Introduction
Malaria remains a major global health challenge, particularly in tropical and subtropical regions. A wide range of antimalarial active pharmaceutical ingredients (APIs) are used in pharmaceutical products, belonging to different chemical classes such as artemisinin derivatives, quinoline derivatives, and antifolates [1].
The investigated compounds included fourteen APIs and three related impurities: artemether, artesunate, atovaquone, chloroquine sulfate, chloroquine phosphate, hydroxychloroquine sulfate, lumefantrine, mefloquine hydrochloride, primaquine diphosphate, proguanil hydrochloride, pyrimethamine, quinidine sulfate, quinine sulfate, sulfadoxine, as well as pyrimethamine impurity B, proguanil hydrochloride impurity B, and chloroquine phosphate impurity C.
Quality control of antimalarial medicines is essential to ensure safety and therapeutic efficacy. However, analytical methods are not always harmonized: some APIs are described in pharmacopoeia monographs using Thin-Layer Chromatography (TLC) identification tests, while others lack standardized TLC procedures [2].
This study aimed to develop a universal HPTLC screening method capable of separating and detecting several antimalarial APIs on a single plate. The method is intended as a rapid screening tool for pharmaceutical quality control, particularly in resource-limited settings. Future work will focus on developing compound-specific HPTLC methods for the detection of impurities and degradation products.
Standard solutions
All APIs were European Pharmacopoeia (Ph. Eur.) reference standards, except for artemether, lumefantrine, and artesunate, which were obtained from MedChemExpress.
Quinine sulfate (CAS 6119-70-6), quinidine sulfate (CAS 6591-63-5), chloroquine sulfate (CAS 6823-83-2), chloroquine phosphate (CAS 50-63-5), chloroquine phosphate impurity C (CAS 86-98-6), hydroxychloroquine sulfate (CAS 747-36-4), primaquine diphosphate (CAS 63-45-6), proguanil hydrochloride (CAS 637-32-1), proguanil hydrochloride impurity B (CAS 106-47-8), sulfadoxine (CAS 2447-57-6), and artesunate (CAS 88495-63-0) were prepared at 0.5 mg/mL in a mixture of methanol, water 4:1 (V/V).
Artemether (CAS 71963-77-4) and lumefantrine (CAS 82186-77-4) were prepared at 0.5 mg/mL in a mixture of dichloromethane, methanol 1:4 (V/V).
Pyrimethamine (CAS 58-14-0), pyrimethamine impurity B (CAS 3275-44-3), mefloquine hydrochloride (CAS 51773-92-3), and atovaquone (CAS 95233-18-4) were prepared at 0.5 mg/mL in methanol.
System Suitability Test (SST): the Universal HPTLC Mix (UHM) was prepared in-house according to [3] or obtained as a ready-to-use solution from Sigma (Prod. No. 91816).
Chromatogram layer
HPTLC plates silica gel 60 F254 (Supelco), 20 × 10 cm, were used.
Sample application
The solutions were applied as bands using the Automatic TLC Sampler (ATS 4), 15 tracks, band length 8.0 mm, distance from the left edge 20.0 mm, track distance 11.4 mm, distance from the lower edge 8.0 mm.
The volumes of the standard solutions were adjusted to achieve appropriate band quality. Proguanil hydrochloride was applied at 1.5 µL. Artemether, lumefantrine, artesunate, pyrimethamine, pyrimethamine impurity B, hydroxychloroquine sulfate, primaquine diphosphate, and chloroquine phosphate were applied at 2.0 µL. Chloroquine sulfate was applied at 3.0 µL. Atovaquone, chloroquine phosphate impurity C, and sulfadoxine were applied at 4.0 µL. Quinine sulfate, quinidine sulfate, and mefloquine hydrochloride were applied at 6.0 µL. Proguanil hydrochloride impurity B was applied at 8.0 µL. The application volume for UHM was 2.0 μL.
Chromatography
Plates were developed in the ADC 2 with chamber saturation for 20 min (with filter paper). Prior to development, the plates were activated at 33% relative humidity for 10 min using a saturated solution of magnesium chloride and pre-conditioned for 5 min. Plates were developed to a migration distance of 70 mm (from the lower edge) and subsequently dried for 5 min.
All APIs and impurities were developed using ammonia 25%, dichloromethane, methanol, toluene 1:60:25:15 (V/V) as the developing solvent.
Post-chromatographic derivatization
After development, for the detection of artemether and artesunate, chemical derivatization was performed using anisaldehyde – sulfuric acid reagent (slowly and carefully mix 170.0 mL of ice-cooled methanol with 20.0 mL of acetic acid and 10.0 mL of sulfuric acid. Allow the mixture to cool to room temperature, then add 1.0 mL of anisaldehyde (p-methoxybenzaldehyde). The plate was sprayed with 3.0 mL of the reagent with the CAMAG Derivatizer (blue nozzle, spraying level 3) and then heated for 3 min at 100 °C on the TLC Plate Heater 3.
Documentation
The plates were documented using the TLC Visualizer 2 under UV 254 nm prior to derivatization, and in white light after derivatization for artemether and artesunate.
Densitometry
Each standard was detected at its maximum of absorption as described in the table below, using TLC Scanner 4 slit dimension 5.00 mm x 0.20 mm, scanning speed 20 mm/s. The UV spectra were recorded from 200 nm to 800 nm using the same slit parameters and a spectrum speed of 20 nm/s.
Results and discussion
Several pharmacopoeia monographs describing antimalarial active pharmaceutical ingredients include thin-layer chromatography (TLC) identification tests.
For example, quinoline derivatives such as quinine sulfate, quinidine sulfate, chloroquine salts, hydroxychloroquine sulfate, mefloquine hydrochloride, and primaquine diphosphate are described in the Ph. Eur. monographs using TLC systems optimized for each compound. Similarly, antifolate antimalarials such as proguanil hydrochloride, pyrimethamine, and sulfadoxine may also be identified using TLC procedures [2].
However, these TLC methods are generally compound-specific and employ different mobile phases and detection conditions. As a result, they are not suitable for the simultaneous analysis of multiple antimalarial APIs on a single chromatographic plate. In addition, some widely used antimalarial drugs, including artemisinin derivatives such as artemether and artesunate, as well as atovaquone and lumefantrine, are not described by TLC identification methods.
Consequently, there is a need for a unified chromatographic approach capable of separating and detecting multiple antimalarial APIs under the same analytical conditions. In this study, an HPTLC method was therefore developed to allow the simultaneous screening of several antimalarial compounds on a single plate.
Selection of the developing solvent
Based on the solvent systems frequently reported in the Ph. Eur. TLC methods, mobile phases composed of combinations of toluene, dichloromethane, methanol, and ammonia solution are commonly used in various ratios.
In the initial test, these solvents were combined in the ratio ammonia (25%), dichloromethane, methanol, toluene 1:60:25:15 (V/V) to provide an appropriate balance between non-polar and polar components while ensuring sufficient migration of basic compounds such as quinoline derivatives.
Under standardized conditions (chamber saturation and plate activation according to Ph. Eur., chapter 2.8.25), this solvent system showed promising potential for the separation of several compounds. However, certain chromatographic issues were observed, including tailing of hydroxychloroquine sulfate and chloroquine sulfate, as well as insufficient separation between pyrimethamine and its impurity, which prompted further optimization of the developing solvent.

HPTLC separation of selected antimalarial APIs on silica gel 60 F254 using ammonia 25%, dichloromethane, methanol, toluene 1:60:25:15 (V/V) as the developing solvent and standard conditions (saturation and activation). Detection under UV 254 nm.
Optimization of the HPTLC method
To enhance the chromatographic performance, other mobile phase modifiers were investigated. Triethylamine and diethylamine were tested as alternative basic additives to replace ammonia. However, neither modifier provided chromatographic behavior better than that achieved with ammonia. Consequently, ammonia remained as the appropriate modifier in the developing solvent. A further improvement was realized by including a pre-conditioning step along with chamber saturation prior to plate development.
This strategy resulted in markedly enhanced chromatographic performance, likely because the pre‑conditioning step exposes the plate to ammonia‑containing vapors from the developing solvent system. The ammonia interacts with and neutralizes acidic silanol groups on the silica surface, thereby reducing surface acidity. This reduction in surface acidity minimizes undesirable secondary interactions with basic analytes.

HPTLC fingerprints of the studied antimalarial APIs and related impurities on silica gel 60 F254 using ammonia 25%, dichloromethane, methanol, toluene 1:60:25:15 (V/V) as the developing solvent and saturation, pre-conditioning and activation. Detection under UV 254 nm prior to derivatization, except for artesunate and artemether, under white light post-derivatization.
Antimalarial APIs and related impurities investigated in this study, including CAS numbers, corresponding Ph. Eur. monographs, RF values obtained using peak profile from image (PPI) at 254 nm and white light (WRT), and the maximum absorption wavelengths (λmax) determined by densitometry.
UV spectra were recorded to discriminate close substances such as primaquine diphosphate and mefloquine hydrochloride (RF 0.253/0.278), or atovaquone and pyrimethamine impurity B (RF 0.632/0.638).

UV/Vis spectra of all APIs and impurities recorded over the wavelength range of 200–800 nm.
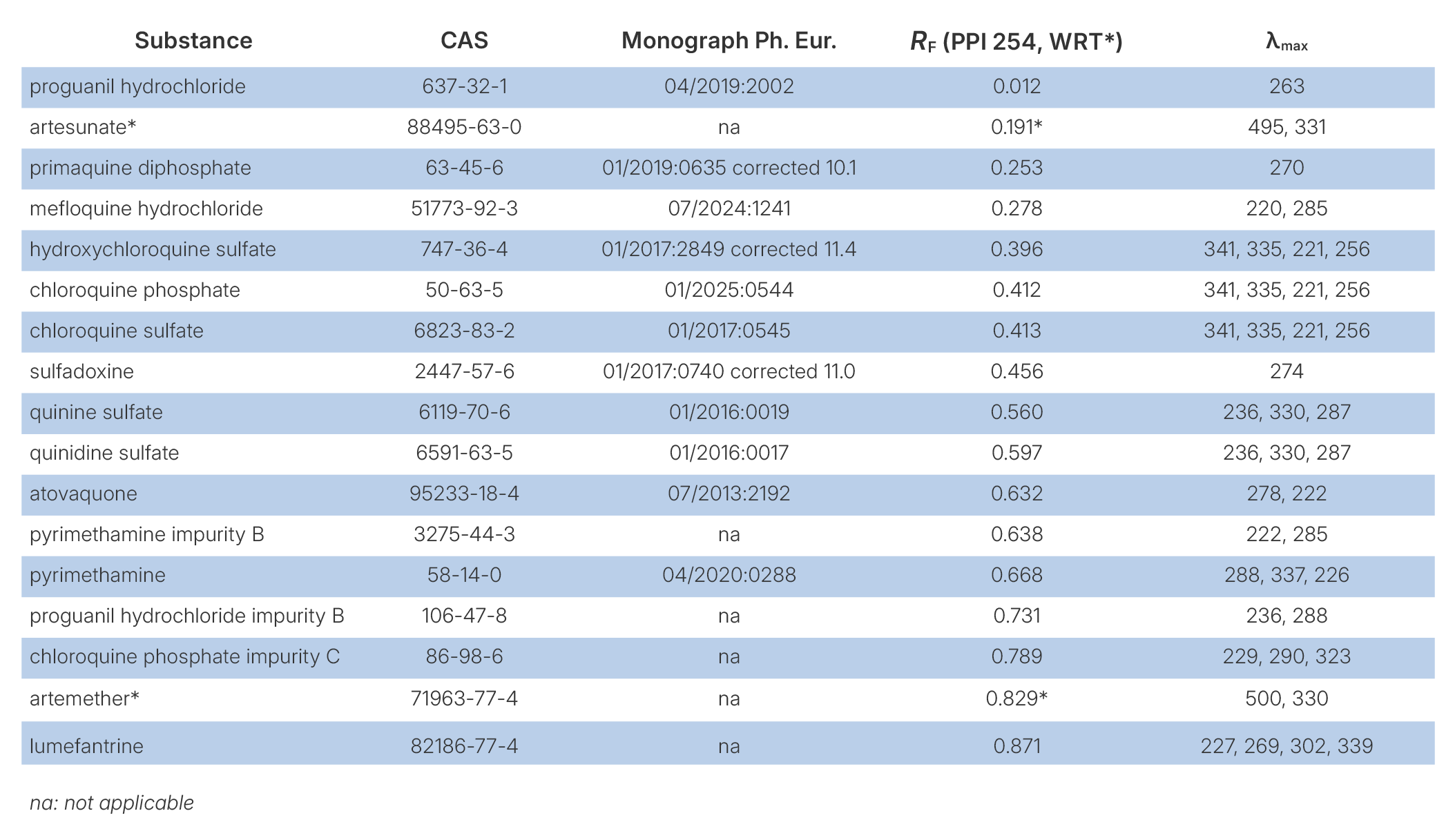
Detection of impurities
The developed HPTLC method allows the detection of the investigated antimalarial APIs as well as several related impurities described in Ph. Eur. monographs. As illustrated in the figure below, compounds such as chloroquine phosphate impurity C, proguanil hydrochloride impurity B, and pyrimethamine impurity B are clearly separated from their corresponding APIs. This demonstrates that the method is suitable not only for the identification of active substances but also for the detection of pharmacopoeia impurities, supporting its applicability for pharmaceutical quality control and screening purposes.
HPTLC separation of selected antimalarial APIs and related Ph. Eur. impurities on silica gel 60 F254 using the selected method.
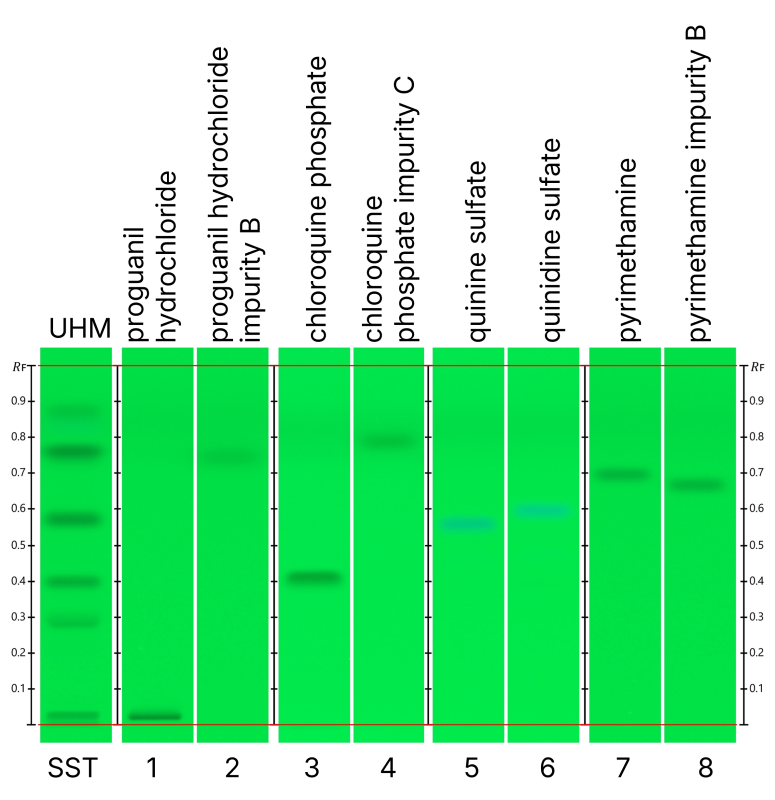
System suitability test (SST)
The method yielded clear and reproducible chromatographic fingerprints. The UHM was applied as SST to verify chromatographic performance prior to analysis. The obtained fingerprint confirmed proper plate development, adequate migration, and consistent separation conditions.
UHM; quenching zones at (n=4):
- RF ~ 0.020 ± 0.004
- RF ~ 0.273 ± 0.010
- RF ~ 0.393 ± 0.009
- RF ~ 0.564 ± 0.008
- RF ~ 0.750 ± 0.008
- RF ~ 0.857 ± 0.011
HPTLC fingerprints of the UHM under UV 254 nm (A) prior to derivatization run on four plates on four different days.
Conclusion
The developed method enables the detection of fourteen APIs and several related impurities, demonstrating its suitability as a rapid screening tool for pharmaceutical quality control, particularly in resource-limited settings.
Future work will focus on the quantitative application of the method and the detection of additional impurities.
Literature
[1] T. M. Belete. Drug Design, Development and Therapy (2020) 14
[2] Ph. Eur. Monographs: 04/2019:2002, 01/2019:0635 corrected 10.1, 07/2024:1241, 01/2017:2849 corrected 11.4, 01/2025:0544, 01/2017:0545, 01/2017:0740 corrected 11.0, 01/2016:0019, 01/2016:0017, 07/2013:2192, 04/2020:0288.
[3] T. K. T. Do et al., J Chrom. A (2021) 1638
Contact:
Dr. Tiên Do, Sonnenmattstrasse 11, 4132 Muttenz, Switzerland, tien.do@camag.com
Tobias Brodda, Sonnenmattstrasse 11, 4132 Muttenz, Switzerland, tobias.brodda@camag.com